Overview
Everything around us is composed of matter. The material of daily use to stuff we can barely see is made up of matter. From the ice cream, we eat in summers to the Sun which causes the seasons, everything is made up of primarily 4 types of matter. They are solid, liquid, gas, and plasma. This classification is done by considering the intermolecular force of attraction between the particles and their arrangement.
“ The state of your psyche defines how you exist in this world.”
States of Matter
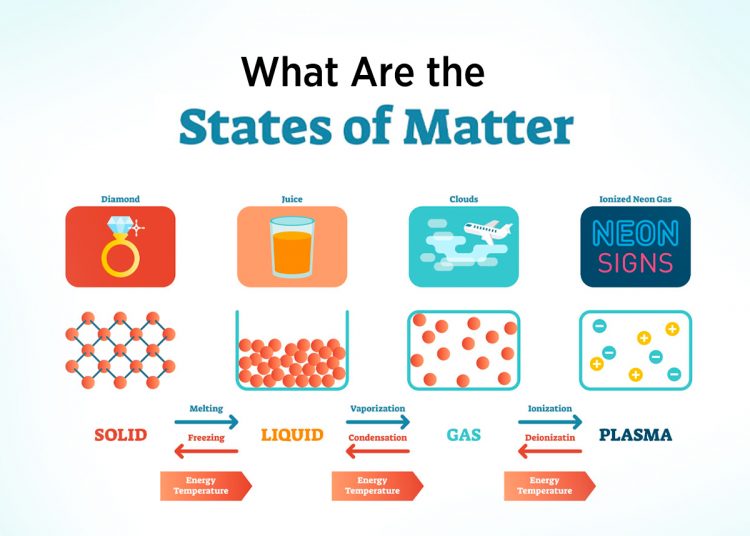
There are 4 fundamental states of matter. Out of which three are naturally occurring and the remaining one is made artificially. Solid, liquid, and gas are the natural states of matter while plasma is man-made. The three natural forms of matter are interconvertible. This means that you can turn a solid into a liquid, or a liquid into gas, and vice versa. The best example can be seen at our homes. The ice is solid in the refrigerator, it becomes liquid at room temperature, and at 100 degrees Celsius, it converts into water vapour (gas).
The matter is anything that has weight and occupies some space. This matter is built of small particles known as atoms. The atoms are made up of smaller subatomic particles known as protons, electrons, and neutrons. These molecules and atoms are held together using chemical energy. This chemical energy is potential energy in disguise. Based on this chemical energy and the arrangement of atoms we will learn the difference between solid, liquid, and gas.
Curiosity Enough: There exists a fifth state of matter which is known as Bose-Einstein condensate. It is also a man-made matter like plasma.
Natural States of Matter
Solid
The chocolate you buy from the confectionery to the chair you sit on, anything where the atoms are packed tightly is known as a solid object. The arrangement of atoms is in such a way that there is no room for movement. The atoms in a solid:
- Are extremely close to each other
- Have fixed position inside the arrangement
- Consists of a very small vibration due to the revolution of electrons
- Cannot travel from their positions
- Have the least amount of kinetic energy out of the three natural states of matter
Due to the above-mentioned behaviour of the particles, the solids have the following properties:
- Solids have a fixed shape. They cannot change their shape until external heat or force is applied to them. This is due to the strong interatomic or intermolecular force of attraction between particles.
- They have constant mass and volume. It is extremely hard to compress solids.
- Solids are immobile. They cannot move on their own.
- The kinetic energy in solids is zero. For this reason, they have the least fluidity.
- Out of the three natural states of matter, solids have the highest density. This is due to the close and tight packing of atoms.
Liquid
Liquids include all the states of matter similar to water. As compared to solids, the particles in a liquid are less tightly packed. The atoms in a liquid can move freely within a limited space. The properties of atoms in liquids are as follows:
- Atoms have considerable space between each other for movement.
- They do not have a definite position inside the arrangement and can travel anywhere inside the liquid.
- In liquids the kinetic energy of atoms is much greater than solids but less than gases.
Due to the aforementioned behaviour of the particles, liquids have the following properties:
- Liquids do not have a definite shape. They can change the shape according to the shape of the solid they are stored in. For instance, water changes its shape from a bottle shape to a glass shape when poured out of the water bottle into a drinking glass.
- The intermolecular force of attraction between two atoms is moderate. For this reason, even liquids are hard to compress.
- Like solids, they also have constant mass and volume.
- The fluidity of liquids is moderate, they can easily travel.
- The density of liquids is less than solids but more than gases.
Gases
The gaseous state of matter forms the entire atmosphere and provides us with oxygen to breathe. The particles are extremely loosely packed in gases. The arrangement of atoms is the most random in this state of matter. Let us look at the properties of gases:
- Particles are extremely far away from one another.
- The atoms have no fixed arrangement at the atomic level.
- Gases travel the fastest and have the highest kinetic energy amongst the three natural states of matter.
Properties of gases
- Like liquids, gases conform to the shape of the container they are kept in.
- The mass and volume are variable. Also, gases are a highly compressible state of matter.
- Solids are the most mobile amongst the three states of matter with the least density.
- Gases also have the highest fluidity due to extreme kinetic energy stored in the particles.
See the figure below to understand the arrangement of particles in each of the states of matter.
The Man-Made States of Matter
Plasma
Plasma is a state of matter whose existence on Earth is still a mystery, but it is present in abundance in outer space. It is possible that 90% of the universe is made up of plasma. Jefferson Laboratory states that it might be the state of matter which made the universe. Examples of plasma are the superheated spheres known as stars. The Sun is also a plasma ball.
Plasma can be defined as the state of matter in which the particles have the highest kinetic energy. It contains highly charged atoms. On Earth, scientists have been successful in making plasma by passing high voltage electricity from noble elements like Helium, Neon, Krypton, etc. On passing electricity, these gases ionise and start to glow. This is used in creating glow and danger signs.
Bose-Einstein Condensate
History: The renowned Indian scientist S.N. Bose, in 1925, sent a paper to Albert Einstein which contained the information about the mathematics of the behaviour of atoms on passing light particles through them. This fascinated Einstein who used this concept to further investigate particles at a quantum level. He observed that when atoms are subjected to a temperature almost equal to absolute zero, they stop behaving like individual particles. The particles lose their identity and behave as a single particle or wave. This state of matter was called a ‘super atom’.
The first BEC was created in 1995 by Eric Cornell and Carl Weiman using lasers and magnets on rubidium at temperatures almost equal to absolute zero. It was observed that at this temperature the molecular motion of the atom comes to a stop. This clumps up the atoms to form the super-atom.
BECs have been useful in studying the dual nature of light and related quantum physics paradoxes. They also exhibit the property of a superfluid. This means they flow without being affected by friction. Many scientists believe that BEC can be the matter which makes the black holes.
Changing the States of Matter
Matter doesn’t stay in a fixed form. Under certain conditions, they may retain a form but if those conditions are altered the matter may change its state. Matter can change from solid → liquid → gas and vice versa. The parameters that affect the state of matter are listed below:
- Temperature: On increasing the temperature the solid converts to liquid. On further heating the liquid converts to gas. Similarly, on decreasing the temperature the gas turns into liquid and the liquid state turns into solid.
- Pressure or Compression: On increasing the pressure the particles are forced to come closer, changing the gas from liquid to solid. Likewise lowering the pressure causes the particles to expand therefore changing the solids from liquids to gases.
Curiosity Enough: When most liquids freeze, they compress, but when water freezes, it expands, making it less dense. Ice may float in water because of this unusual property. The icebergs in Antarctica are the best example of this property.
The processes of changing the states of matters are given in detail below:
Sublimation
The direct transformation of solid state to gaseous state is termed as sublimation. This is achieved by increasing the temperature of a solid to immediately reach its boiling point. An alternative way is also used which is called ‘freeze-drying’. In this process, the water molecules from the solids are removed by cooling them in a vacuum. The water is removed for the solid to not change to liquid first. The elements which transform from solid to gas are known as volatile substances. Some of the common volatile substances are Sodium, Potassium, dry ice, etc.
Vaporisation
The method of conversion of liquid into the gaseous state is known as vaporisation. This can be achieved by either evaporating or boiling the liquid to its boiling point. The boiling point is the temperature at which a liquid starts to turn into gas. This can vary from liquid to liquid. The concept behind vaporisation is that a liquid’s particles are constantly moving and regularly collide with one another. When enough energy is imparted to particles at the surface, they can be knocked away completely from the specimen as a free gas molecule
Condensation
Condensation is the phenomenon in which the gases particles change to liquid. In this process when the gaseous molecules are cooled down they gather together to again form liquid molecules. Condensation occurs naturally in the water cycle. When the gases are cooled they lose their kinetic energy. Since they cannot move further the gaseous molecules accumulate together to give us a liquid.
Pressure is also an important factor in condensation. Condensation can speed up with the increase in pressure and vice versa.
Frequently Asked Questions
1) Is it possible for matter to exist in two states at the same time?
Ans. Pressure can change the state of matter. Certain things vary a lot, but they usually only exist in two states and need managerial and technological assistance to get through all three stages. Water exists as solid, liquid and gas. It is the only known substance to exhibit all three states of matter.
2) What is the composition of matter?
Ans. Small particles make up the matter as well. Atoms are the makers of matter. Atoms are invisible because they are so little. The majority of atoms combine to form matter.
3) Can we say that light is a matter?
Ans. Light is a kind of energy, not matter. To be precise, light is electromagnetic radiation. At times light behaves as a particle (like solid, liquid, or gas) but other times behaves as waves. Thus it is impossible to understand the composition of light.
4) Are there more states of matter?
Ans. Yes! There exist 2 more states besides the 5 states of the matter covered in the above article, which is solid, liquid, gas, plasma, and Bose-Einstein Condensate. They are Quark-Gluon Plasma and Fermionic Condensate. There may exist many more states of matter which are present in superheated or supercooled conditions. These are beyond human reach right now.
5) Are states of matter important for exams?
Ans. Somewhat yes! The states of matter are not that important from an examination point of view. Very few times questions from this topic have appeared in competitive exams. But it is important to know the concepts of this chapter for general knowledge.