In the realm of thermodynamics, the Carnot Cycle stands as a foundational concept that sheds light on the theoretical limits of the efficiency of heat engines. Developed by French engineer Sadi Carnot in 1824, this cycle serves as a benchmark for evaluating the maximum possible efficiency that any heat engine, operating between two temperature reservoirs, can achieve. The Carnot Cycle is not only a crucial theoretical framework but also provides insights into the practical design and performance of various energy conversion systems.
The Four Stages of the Carnot Cycle:
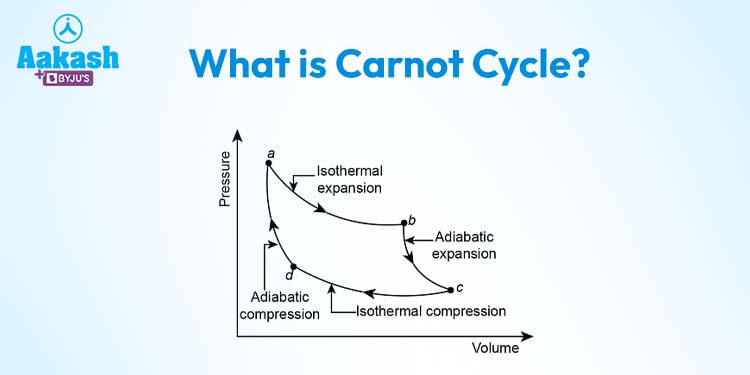
The Carnot Cycle consists of four distinct stages, each involving specific thermodynamic processes: isothermal expansion, adiabatic expansion, isothermal compression, and adiabatic compression.
Isothermal Expansion (Heat Absorption at High Temperature): In this phase, the working substance (often a gas) undergoes an isothermal (constant temperature) expansion while absorbing heat from the high-temperature reservoir. During this expansion, the gas does work on the surroundings while its temperature remains constant. This process ensures that the gas remains in thermal equilibrium with the high-temperature reservoir.
Adiabatic Expansion (Reversible Adiabatic Process): Following the isothermal expansion, the gas undergoes an adiabatic expansion, during which it expands without exchanging heat with its surroundings. This leads to a decrease in both the pressure and temperature of the gas.
Isothermal Compression (Heat Rejection at Low Temperature): In this phase, the gas is compressed isothermally, releasing heat to the low-temperature reservoir. The gas performs work on the surroundings as it is compressed back to its original state. The temperature of the gas remains constant throughout this process, and it remains in thermal equilibrium with the low-temperature reservoir.
Adiabatic Compression (Reversible Adiabatic Process): The final stage involves adiabatic compression, during which the gas is compressed further without any heat exchange. This process results in an increase in both the pressure and temperature of the gas, returning it to its initial state and completing the cycle.
The Carnot Efficiency:
The efficiency of a heat engine is defined as the ratio of useful work output to the heat input. For the Carnot Cycle, the efficiency is remarkably high due to its idealized nature and the principles of reversibility.
Practical Implications and Limitations:
While the Carnot Cycle represents an idealized model, real-world heat engines can never achieve the Carnot efficiency due to various irreversibilities, losses, and practical constraints. Nonetheless, the Carnot Cycle serves as an essential benchmark for engineers and scientists, guiding them to design more efficient heat engines by minimizing losses and optimizing operating conditions.
In essence, the Carnot Cycle not only deepens our understanding of thermodynamics but also offers a fundamental framework for assessing the upper limits of efficiency in energy conversion systems. Its enduring significance continues to shape the development of technologies that harness heat energy for a wide range of applications, from power generation to refrigeration and beyond.
FAQs About the Carnot Cycle:
1. What is the Carnot Cycle?
The Carnot Cycle is a theoretical thermodynamic cycle that describes the idealized operation of a heat engine between two temperature reservoirs. It consists of four stages: isothermal expansion, adiabatic expansion, isothermal compression, and adiabatic compression. The cycle serves as a benchmark for determining the maximum possible efficiency of heat engines.
2. What is the significance of the Carnot Cycle?
The Carnot Cycle is significant because it establishes the upper limit of efficiency that any heat engine can achieve when operating between two temperature extremes. It provides a reference point for comparing the actual performance of real-world heat engines and offers valuable insights for improving their efficiency by minimizing losses and optimizing processes.
3. Can real heat engines achieve Carnot efficiency?
No, real heat engines cannot achieve the Carnot efficiency due to various practical limitations and irreversibilities. The Carnot Cycle assumes perfectly reversible processes, while real-world engines experience friction, heat losses, and other inefficiencies that prevent them from reaching the ideal efficiency. Nonetheless, engineers strive to get as close to Carnot’s efficiency as possible through design improvements.
4. What practical applications are influenced by the Carnot Cycle?
The principles of the Carnot Cycle have applications in various fields, such as power generation, refrigeration, and heating systems. Power plants, including steam turbines and internal combustion engines, draw insights from the cycle to optimize energy conversion. Refrigeration and heat pump systems use these principles to enhance cooling and heating processes, making them more energy-efficient.