Atomic radius is a fundamental concept in chemistry that describes the size of an atom. It is a critical property that influences various atomic and chemical behaviors. Understanding atomic radius is essential for comprehending periodic trends, chemical bonding, and the overall behavior of elements in the periodic table. This article explores the definition of atomic radius, the factors affecting it, and its significance in the world of chemistry.
Definition of Atomic Radius
The atomic radius of an atom refers to the distance from the nucleus to the outermost electron’s orbital boundary. It is important to note that atoms do not have a sharp or well-defined boundary; rather, they possess a fuzzy electron cloud that extends outward from the nucleus. Thus, atomic radius is an average value, often represented by the symbol “r,” and is typically measured in picometers (pm) or angstroms (Å).
Factors Affecting Atomic Radius
Nuclear Charge:
The number of protons in the atom’s nucleus (atomic number) plays a significant role in determining the atomic radius. As the nuclear charge increases, the positive charge in the nucleus pulls the electrons closer, reducing the atomic radius. Conversely, a decrease in nuclear charge leads to a larger atomic radius.
Electron Shells:
The number of electron shells or energy levels also affects atomic radius. As we move down a group in the periodic table, additional electron shells are added, resulting in a larger atomic radius due to the increased distance between the nucleus and the outermost electrons.
Electronegativity:
Electronegativity is an atom’s tendency to attract electrons toward itself when involved in a chemical bond. Higher electronegativity typically leads to a smaller atomic radius since the atom’s strong pull draws the electrons closer to the nucleus.
Electron-Electron Repulsion:
Within an electron shell, electrons repel each other due to their negative charges. This electron-electron repulsion causes the electron cloud to spread out, resulting in a larger atomic radius.
Significance of Atomic Radius
Periodic Trends:
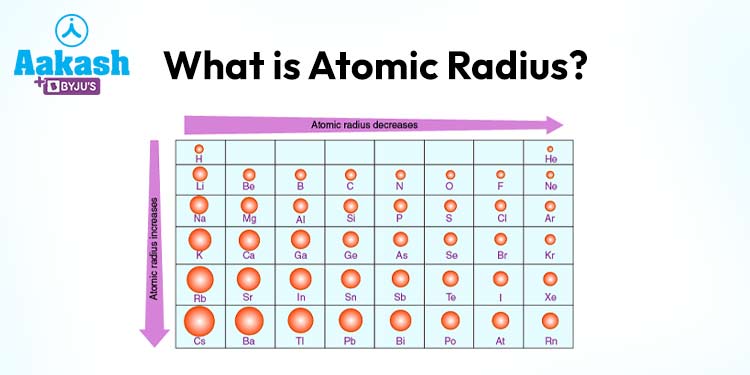
Atomic radius is a key component of periodic trends. As you move across a period from left to right in the periodic table, the atomic radius generally decreases due to the increasing nuclear charge, even though the number of electron shells remains constant. Conversely, moving down a group results in an increase in atomic radius due to the addition of new electron shells.
Chemical Bonding:
Atomic radius significantly influences the nature of chemical bonding. When atoms with large differences in atomic radii bond, such as a metal and a nonmetal, ionic bonding often occurs. In contrast, atoms with similar atomic radii tend to form covalent bonds, sharing electrons to achieve stability.
Atomic Volume:
The atomic radius contributes to the overall volume of an element in its solid state. Elements with larger atomic radii generally have higher atomic volumes, affecting their density and other physical properties.
Conclusion:
In conclusion, atomic radius is a crucial property that describes the size of an atom and has a profound impact on various chemical and physical behaviors. The factors influencing atomic radius, such as nuclear charge, electron shells, electronegativity, and electron-electron repulsion, all play a vital role in determining the overall behavior of elements in the periodic table. Understanding atomic radius is fundamental for comprehending the complexities of chemical bonding, periodic trends, and the nature of elements.
FAQs about Atomic Radius
How is Atomic Radius measured?
Atomic radius is challenging to measure directly due to the absence of well-defined atomic boundaries. However, scientists use various theoretical and experimental methods to estimate atomic radii. One common approach is to measure the distance between the nuclei of two adjacent atoms in a solid lattice and divide it by two to obtain an approximate atomic radius.
What factors affect Atomic Radius?
The atomic radius of an element is influenced by two main factors: the number of electron shells and the effective nuclear charge. As you move across a period (horizontal row) in the periodic table, the atomic radius generally decreases due to an increase in effective nuclear charge, pulling the electrons closer to the nucleus. Conversely, as you move down a group (vertical column), the atomic radius generally increases because new electron shells are added, leading to a larger electron cloud.
How does Atomic Radius relate to the reactivity of elements?
Atomic radius significantly influences an element's reactivity. Generally, larger atoms with larger atomic radii tend to be more reactive because their outermost electrons are farther from the nucleus and are less tightly held. These electrons are more easily lost or shared during chemical reactions, making the element more reactive.
How does the concept of Atomic Radius apply to ions?
When an atom gains or loses electrons, it forms an ion. The atomic radius of an ion can differ significantly from that of its parent atom. For cations (positively charged ions), the atomic radius decreases compared to the neutral atom because fewer electron shells remain. Conversely, anions (negatively charged ions) have a larger atomic radius than the parent atom due to the addition of extra electrons, leading to increased electron-electron repulsions and a larger electron cloud.
Why does Helium have a smaller Atomic Radius than Hydrogen?
Helium and hydrogen are both in the first period of the periodic table and contain only one electron shell. However, helium has a smaller atomic radius than hydrogen. This difference is attributed to the effective nuclear charge. Helium's nucleus has two protons, while hydrogen has only one. With the same electron shell, helium's nucleus exerts a stronger pull on the single electron, making its atomic radius smaller compared to hydrogen's single proton nucleus.