Electricity is a fundamental force of nature that plays a crucial role in our daily lives. When electric current flows through a conductive material, it generates various effects, one of which is the “Chemical Effect of Electric Current.” This phenomenon is the basis for a wide range of applications, including electroplating, electrolysis, and electrochemical cells.
Understanding the Chemical Effect:
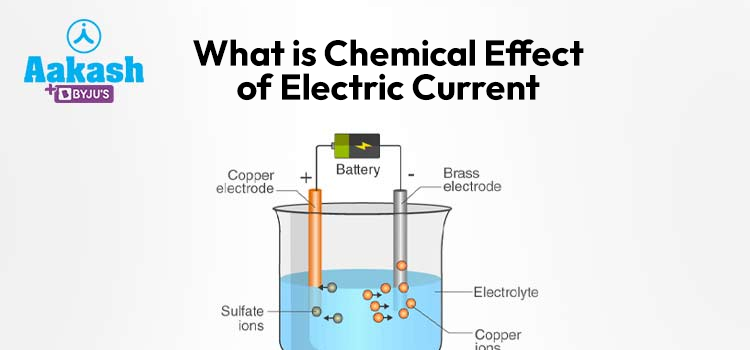
The chemical effect of electric current occurs when an electric current passes through an electrolyte, a substance that can conduct electricity. Electrolytes are usually solutions of acids, bases, or salts in water or molten ionic compounds. When an electric potential difference is applied across the electrolyte, positive and negative ions present in the solution begin to migrate toward the oppositely charged electrodes.
Electrodes:
In any electrolytic setup, two electrodes are immersed in the electrolyte. One is the anode, which attracts negatively charged ions (anions), and the other is the cathode, which attracts positively charged ions (cations).
Electrolysis:
Electrolysis is a significant application of the chemical effect of electric current. During electrolysis, the electric current forces a chemical reaction to occur at the electrodes. At the anode, oxidation takes place, where negatively charged ions lose electrons and become neutral atoms or molecules. At the cathode, reduction occurs, where positively charged ions gain electrons and are transformed into neutral atoms or molecules.
Electroplating:
Electroplating is another practical application of the chemical effect of electric current. It involves coating a metal object with a thin layer of another metal through electrolysis. The metal object to be plated is made the cathode, and the metal to be deposited is used as the anode. As the electric current passes through the electrolyte, metal ions from the anode are reduced and deposited onto the cathode, creating a uniform and adherent metal coating.
Electrochemical Cells:
Electrochemical cells, such as batteries and fuel cells, are devices that convert chemical energy into electrical energy or vice versa. In these cells, chemical reactions occur at the electrodes when current flows through the cell. During discharge (use of the battery), the chemical reactions produce electricity, and during charging, electricity is used to drive the chemical reactions, storing energy for later use.
Corrosion:
While the chemical effect of electric current finds numerous constructive applications, it can also lead to destructive consequences. Corrosion is one such example. When metals are exposed to an electrolyte (e.g., moisture or salt water) and a potential difference exists, the metal can undergo corrosion, causing deterioration and damage over time.
Conclusion:
The chemical effect of electric current is a fascinating aspect of electricity, with wide-ranging applications in various industries and our everyday lives. Understanding and harnessing this phenomenon have led to advancements in electroplating, electrolysis, and the development of efficient electrochemical cells. By using electric current to drive chemical reactions, we can achieve numerous practical and innovative outcomes that contribute to technological progress and the betterment of society. However, it is essential to consider the potential detrimental effects, such as corrosion, and take necessary measures to mitigate them for sustainable and responsible usage.
FAQs – The Chemical Effect of Electric Current
1. What is the chemical effect of electric current?
The chemical effect of electric current refers to the phenomenon in which an electric current passing through an electrolyte causes a chemical reaction at the electrodes. This results in the transformation of ions into neutral atoms or molecules, leading to various applications such as electrolysis, electroplating, and the operation of electrochemical cells.
2. How does electrolysis work, and what are its applications?
Electrolysis is a process that uses the chemical effect of electric current to drive a non-spontaneous chemical reaction. When an electric potential is applied across an electrolyte, the positive ions move toward the cathode and undergo reduction, while the negative ions move toward the anode and undergo oxidation. Electrolysis finds applications in industries such as metal extraction, electroplating, and the production of certain chemicals.
3. What is electroplating, and how does it use the chemical effect of electric current?
Electroplating is a technique used to coat a metal object with a thin layer of another metal. In this process, the metal object to be plated is made the cathode, and the metal to be deposited is used as the anode. As electric current passes through the electrolyte, metal ions from the anode are reduced and deposited onto the cathode, forming a uniform and adherent metal coating. Electroplating is widely used in industries like jewelry manufacturing and electronics.
4. How do electrochemical cells work, and what are their applications?
Electrochemical cells are devices that convert chemical energy into electrical energy or vice versa. These cells involve chemical reactions occurring at the electrodes when an electric current flows through them. During discharge, the chemical reactions produce electricity, and during charging, electricity is used to drive the chemical reactions, storing energy for later use. Batteries and fuel cells are examples of electrochemical cells with applications in portable electronics, electric vehicles, and renewable energy systems.
5. Can the chemical effect of electric current have negative consequences?
Yes, the chemical effect of electric current can lead to negative consequences, such as corrosion. When metals are exposed to an electrolyte and an electric potential difference exists, the metal can corrode, causing deterioration and damage over time. To mitigate such effects, protective coatings, sacrificial anodes, and other preventive measures are employed to ensure the longevity and reliability of metal structures and devices.