Picture a water droplet resting delicately on a leaf, or the way insects like water striders seem to skate effortlessly across the surface of a pond. These intriguing phenomena are governed by a fundamental property of liquids known as surface tension. Surface tension is a captivating concept that unveils the subtle interactions occurring at the interface between a liquid and its surroundings.
Defining Surface Tension:
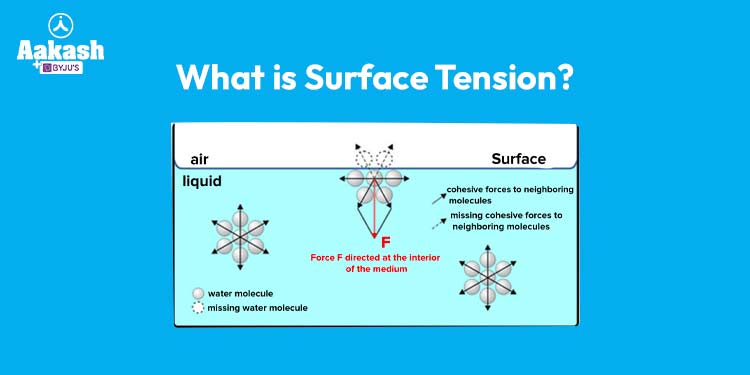
Surface tension refers to the cohesive forces that bind the molecules of a liquid together at its exposed surface. It’s a result of the unbalanced molecular forces acting on the molecules at the surface of the liquid, causing them to behave as if they were under an invisible, elastic membrane. This phenomenon gives rise to some remarkable behaviors and effects that have intrigued scientists, engineers, and curious minds for centuries.
The Role of Intermolecular Forces:
At the heart of surface tension are the intermolecular forces that determine how molecules interact with each other. These forces include hydrogen bonding, dipole-dipole interactions, and van der Waals forces. In a liquid, molecules are attracted to each other from all directions, creating a net inward force at the surface. This cohesive force creates a layer of molecules that is more tightly bound compared to the molecules in the bulk of the liquid.
Effects and Phenomena:
Surface tension is responsible for a range of fascinating phenomena and everyday observations:
Capillary Action: This is the ability of a liquid to flow against gravity in a narrow space, such as a thin tube or the fibers of a paper towel. Capillary action is the result of the adhesive forces between the liquid and the solid surfaces coupled with the cohesive forces within the liquid.
Droplet Formation: Surface tension plays a vital role in forming droplets, whether it’s raindrops falling from the sky or the spherical shape of a water droplet on a surface. The forces at play strive to minimize the surface area, leading to the spherical shape that minimizes the ratio of surface area to volume.
Meniscus Formation: When you look at a graduated cylinder or a glass of water, you’ll notice a curved surface at the edges where the liquid meets the container. This is known as the meniscus, and it’s a result of surface tension and adhesive forces between the liquid and the container.
Floating and Buoyancy: Small insects like water striders can seemingly defy gravity and float on the surface of water due to surface tension. Additionally, surface tension can influence buoyancy, affecting whether an object will sink or float on a liquid.
Practical Applications:
Surface tension’s unique properties have practical applications in various fields:
Industrial Processes: Surface tension is crucial in processes like painting, printing, and coating, where it affects how liquids spread and adhere to surfaces.
Biological Systems: Surface tension plays a role in lung function, aiding in the exchange of gases during respiration. It also contributes to the capillary action that helps plants transport water from roots to leaves.
Materials Science: Researchers use surface tension to understand and manipulate the behavior of liquids on surfaces, which is important in the development of materials like superhydrophobic coatings.
In conclusion, surface tension is a captivating concept that reveals the intricate interplay of forces at the boundary between liquids and their surroundings. From the mesmerizing behavior of water droplets to the practical applications in various fields, surface tension offers a glimpse into the hidden forces that shape our world at the molecular level.
FAQs About Surface Tension
1. What is surface tension?
Surface tension is a property of liquids arising from the cohesive forces between molecules at the liquid’s surface. It causes the surface molecules to behave as if they were under an invisible elastic film, resulting in unique behaviors and phenomena.
2. How does surface tension work?
Surface tension is a consequence of intermolecular forces like hydrogen bonding, dipole-dipole interactions, and van der Waals forces. These forces create an imbalance, causing molecules at the liquid’s surface to be more strongly attracted to each other than to the surrounding medium.
3. What are some everyday examples of surface tension?
Several phenomena are influenced by surface tension, including droplet formation (raindrops and dew), capillary action (water rising in narrow tubes), and the spherical shape of water droplets. It also enables small insects like water striders to float on the water’s surface.
4. How does surface tension affect objects in liquids?
Surface tension can cause objects to either sink or float in liquids. It affects buoyancy and is responsible for the meniscus formation in containers. Objects denser than the liquid might sink due to gravitational forces, while those less dense may be supported by surface tension and appear to “float.”
5. What are the practical applications of surface tension?
Surface tension has various real-world applications. It influences the way liquids spread and adheres in industrial processes like painting and coating. In biology, it aids in lung function and helps plants transport water. In materials science, surface tension is harnessed to design specialized coatings with superhydrophobic properties.
6. How is surface tension measured?
Surface tension is typically measured in units of force per unit length, such as newtons per meter (N/m) or dynes per centimeter (dyn/cm). Common methods for measuring surface tension include the capillary rise method, the drop weight method, and the Wilhelmy plate method, among others.