Surface energy is a fundamental concept in physics and materials science that describes the energy associated with the surface of a material. It plays a crucial role in shaping the behavior of materials at interfaces, influencing phenomena such as wetting, adhesion, and even the stability of structures. To comprehend surface energy, one must delve into the underlying forces and interactions that govern the behavior of particles at the surface.
Defining Surface Energy:
Surface energy, also known as interfacial energy or surface tension, is the excess energy present at the surface of a material compared to its bulk. It arises due to the imbalance of cohesive forces between atoms or molecules within the bulk of the material and the surface. This discrepancy in forces leads to the surface acting as if it were under tension, trying to minimize its area and attain a state of lower energy.
Intermolecular Forces and Surface Energy:
At the heart of surface energy are intermolecular forces, the attractive and repulsive forces between molecules. These forces include:
Van der Waals Forces: These weak, short-range forces emerge from temporary fluctuations in electron distributions, creating temporary dipoles that induce similar dipoles in neighboring molecules. While individually weak, these forces collectively contribute to the overall cohesion of the material.
Electrostatic Forces: Charged particles within molecules can lead to electrostatic attractions or repulsions between neighboring molecules. These forces are particularly relevant when dealing with polar molecules.
Hydrogen Bonding: In molecules with hydrogen atoms bonded to highly electronegative atoms (like oxygen, nitrogen, or fluorine), hydrogen bonds form. These bonds are stronger than typical van der Waals interactions and significantly impact the surface properties of a material.
Ionic Interactions: Materials with ionic bonding exhibit strong electrostatic attractions between oppositely charged ions, further influencing surface behavior.
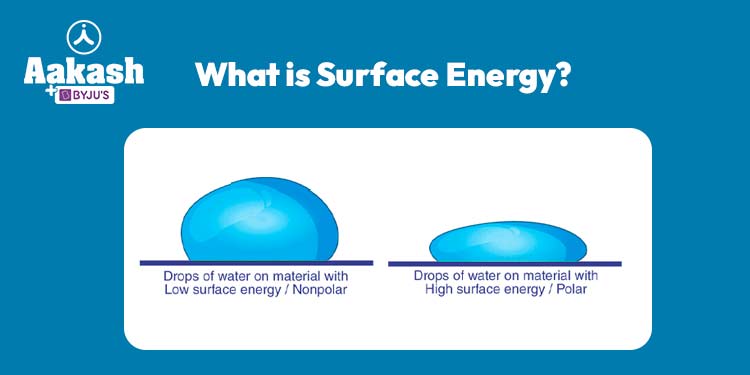
Contact Angle and Wetting:
Surface energy is closely linked to the phenomenon of wetting, which describes how a liquid spreads across a solid surface. The degree to which a liquid wets a surface can be quantified by the contact angle formed at the liquid-solid interface. A low contact angle indicates strong wetting and suggests that the liquid spreads out over the surface, while a high contact angle indicates poor wetting, causing the liquid to bead up.
Adhesion and Cohesion:
Surface energy also plays a pivotal role in adhesion—the bonding of two different materials at their interface. Adhesion is influenced by the compatibility of surface energies between the two materials. If the surface energies are similar, adhesion is likely to be strong. On the other hand, materials with dissimilar surface energies tend to repel each other, leading to poor adhesion.
Applications of Surface Energy:
Coatings and Paints: Surface energy considerations are critical in developing coatings and paints that adhere well to different substrates. A proper understanding of surface energy helps in formulating paints that can wet and adhere effectively to a variety of surfaces.
Biomedical Devices: Surface energy influences the behavior of materials used in medical implants and devices. Implant surfaces with controlled surface energy can encourage better integration with surrounding tissues.
Microfluidics: In microfluidic devices, precise control of surface energy is necessary to manipulate and control the flow of small amounts of fluids within microchannels.
Nanotechnology: Surface energy becomes particularly significant at the nanoscale, influencing nanoparticle interactions, self-assembly, and nanomaterial properties.
In summary, surface energy is a complex yet essential concept that underlies a wide range of natural and engineered phenomena. It results from the interplay of various intermolecular forces and significantly influences how materials interact with each other and their environment. Understanding and harnessing surface energy enables us to design innovative materials and technologies with tailored properties for diverse applications.
Frequently Asked Questions (FAQs) About Surface Energy
1. What is surface energy?
Surface energy refers to the excess energy present at the surface of a material compared to its bulk. It arises due to the imbalance of cohesive forces between molecules in the bulk and those at the surface. This energy imbalance causes the surface to act as if it’s under tension, striving to minimize its area and attain a state of lower energy.
2. How is surface energy related to intermolecular forces?
Surface energy is closely tied to intermolecular forces—the attractive and repulsive forces between molecules. These forces include van der Waals forces, electrostatic forces, hydrogen bonding, and ionic interactions. The strength and nature of these forces influence the surface properties of a material and its interactions with other substances.
3. What is the role of surface energy in wetting and contact angle?
Surface energy plays a significant role in wetting, which describes how a liquid spreads over a solid surface. The contact angle formed at the liquid-solid interface indicates the degree of wetting. A low contact angle suggests strong wetting, while a high angle indicates poor wetting. Surface energy determines the balance between adhesive and cohesive forces at the interface.
4. How does surface energy affect adhesion?
Surface energy is a crucial factor in adhesion—the bonding of two different materials at their interface. Materials with similar surface energies tend to adhere well, while materials with dissimilar surface energies repel each other. A proper understanding of surface energy helps in designing materials and coatings for strong adhesion in various applications.
5. What are some practical applications of surface energy considerations?
Surface energy plays a vital role in several applications:
Coatings and Paints: Formulating coatings and paints that adhere effectively to different surfaces.
Biomedical Devices: Enhancing the integration of medical implants with surrounding tissues.
Microfluidics: Manipulating fluid flow in microfluidic devices through controlled surface energy.
Nanotechnology: Influencing nanoparticle interactions and self-assembly for nanomaterial design.
Materials Engineering: Tailoring material properties for specific uses by manipulating surface energy.
These applications demonstrate the significance of understanding and harnessing surface energy in various fields of science and engineering.