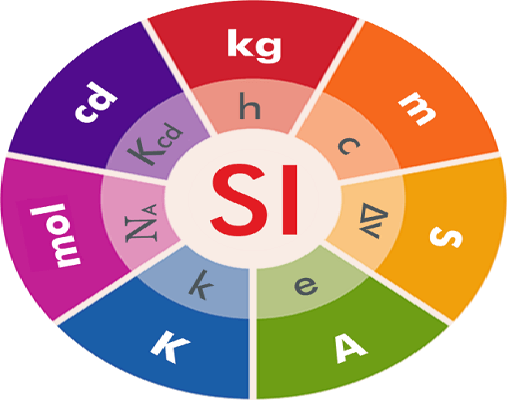
What is SI Unit?
The International System of Units, abbreviated as SI (from the French “Système International d’Unités”), is the modern metric system of measurement used worldwide. It provides a standard framework for measuring various physical quantities in a consistent and universally understood manner. The SI system is based on a set of seven base units, from which all other units are derived.
Here are the seven base SI units:
Meter (m): The meter is the base unit of length. It is defined as the distance traveled by light in a vacuum during a specific time interval. This definition provides a precise and constant value for the meter.
Kilogram (kg): The kilogram is the base unit of mass. Historically, it was defined as the mass of a specific platinum-iridium alloy cylinder known as the International Prototype of the Kilogram (IPK). However, in recent years, a more accurate definition based on fundamental constants has been adopted.
Second (s): The second is the base unit of time. It is defined as the duration of 9,192,631,770 oscillations of a specific caesium-133 atom when it transitions between two energy states.
Ampere (A): The ampere is the base unit of electric current. It is defined as the constant current that, if maintained in two straight parallel conductors of infinite length and negligible cross-section, placed one meter apart in a vacuum, would produce a specific force between them.
Kelvin (K): The kelvin is the base unit of temperature. It is defined based on the Boltzmann constant, which relates temperature to the average kinetic energy of particles in a system.
Mole (mol): The mole is the base unit of the amount of substance. It is defined as the amount of substance that contains as many elementary entities (atoms, molecules, ions, etc.) as there are atoms in exactly 0.012 kilograms of carbon-12.
Candela (cd): The candela is the base unit of luminous intensity. It is defined based on the radiation emitted by a specific isotropic light source.
| Sl. No. | Name of the Quantity | SI Unit | SI Unit Symbol |
| 1. | Length (l) | Meter | m |
| 2. | Mass (M) | Kilogram | kg |
| 3. | Time (T) | Second | s |
| 4. | Electric current (I) | Ampere | A |
| 5. | Thermodynamic temperature (Θ) | Kelvin | K |
| 6. | Amount of substance (N) | Mole | mol |
| 7. | Luminous intensity (J) | Candela | cd |
In addition to these base units, the SI system also includes derived units that are combinations of the base units. For example, the unit of speed is derived from the base units of distance (meter) and time (second), resulting in meters per second (m/s).
The SI system provides a coherent and standardized approach to measurement, facilitating accurate and consistent communication across scientific disciplines and international borders. It allows scientists, engineers, and individuals worldwide to express and compare measurements with ease, promoting a common understanding of physical quantities.
FAQs
The purpose of the SI unit system is to provide a standardized and universally accepted framework for measuring physical quantities. It allows for consistent communication and understanding of measurements across different scientific disciplines and countries. Base units in the SI system serve as the fundamental building blocks for measuring different physical quantities. They provide a starting point for deriving and defining other units. The use of base units allows for greater coherence and consistency in measurement across various fields of science and technology. While the SI system provides a unified approach to measurement, there are some exceptions and variations. Certain specialized fields may use additional units or specific conventions, but these are typically built upon the foundation of the SI system. Additionally, some non-SI units, such as minutes and hours, are widely used alongside SI units for time measurement. Converting between SI units is typically straightforward as it involves multiplying or dividing by factors of ten. For example, to convert from meters (m) to kilometers (km), you divide the value by 1000. Similarly, to convert from kilograms (kg) to grams (g), you multiply the value by 1000. Conversion factors for various SI units can be found in conversion tables or by using online unit conversion tools.What is the purpose of the SI unit system?
Why are there base units in the SI system?
Are there any exceptions or variations within the SI unit system?
How can I convert between different SI units?