Bragg’s Law stands as one of the cornerstones in the field of X-ray crystallography, a scientific discipline that has revolutionized our understanding of the molecular and atomic structures of various materials. Proposed by British physicists William Henry Bragg and his son William Lawrence Bragg in 1913, this law has played a pivotal role in unlocking the mysteries hidden within the intricate lattice structures of crystals.
The Essence of Bragg’s Law: Interference Unveiling Structure
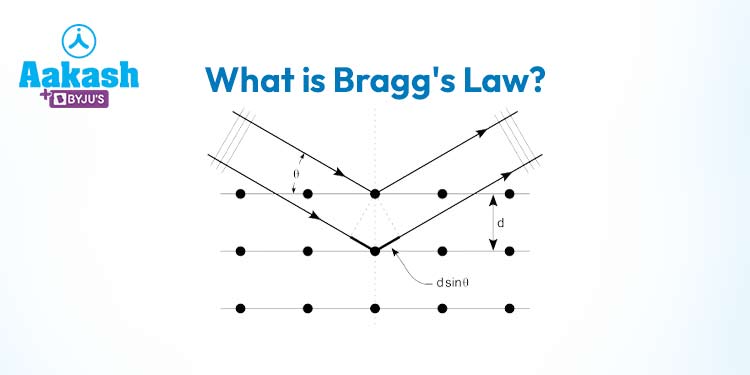
At its core, Bragg’s Law revolves around the phenomenon of X-ray diffraction – the scattering of X-rays as they pass through a crystal lattice. This scattering process gives rise to patterns that contain valuable information about the arrangement of atoms within the crystal. Bragg’s Law provides a quantitative explanation for these diffraction patterns, linking the angles at which the X-rays are scattered with the distances between atomic planes within the crystal.
Constructive interference occurs when the waves’ troughs and crests align, resulting in a stronger, amplified wave. Bragg’s Law helps us determine the angles at which this constructive interference occurs. When the X-rays are incident at these angles, they interfere constructively after bouncing off the crystal planes, resulting in enhanced diffraction.
The Real-World Impact: Unraveling Molecular Structures
Bragg’s Law has had a profound impact on various scientific disciplines. One of its most notable applications is in the field of X-ray crystallography, where it has been used to decipher the three-dimensional structures of molecules, such as proteins, enzymes, and other complex organic compounds. This technique has revolutionized fields like biology and chemistry, enabling scientists to understand the function of these molecules by visualizing their atomic arrangements.
By analyzing the diffraction patterns produced when X-rays pass through crystals, researchers can mathematically deduce the spatial arrangements of atoms within the crystal. This knowledge, in turn, provides insights into the molecule’s properties, behavior, and interactions with other molecules. X-ray crystallography has been instrumental in drug discovery, material science, and many other areas.
In Conclusion: Unraveling Nature’s Puzzle
Bragg’s Law, a result of ingenious thinking by father and son, stands as an exemplar of the profound connection between physics and its applications in understanding the intricacies of the natural world. Its contribution to the development of X-ray crystallography has transformed our ability to visualize and comprehend the hidden architecture of matter, unlocking doors to innovative applications across diverse scientific disciplines. As technology advances, Bragg’s Law continues to be a cornerstone in our quest to unravel the fundamental structures that underlie the materials shaping our universe.
Frequently Asked Questions (FAQs) about Bragg’s Law
1. What is Bragg’s Law and why is it significant?
Bragg’s Law is a fundamental principle in X-ray crystallography, a scientific technique used to determine the atomic and molecular structures of crystalline materials. It explains how X-rays are diffracted by crystal lattices and provides a way to calculate the angles at which diffraction occurs. This law has revolutionized fields like chemistry and biology by enabling researchers to visualize complex molecules’ structures, leading to breakthroughs in drug development and materials science.
2. How does Bragg’s Law relate to X-ray diffraction?
Bragg’s Law is intimately connected to X-ray diffraction, a phenomenon that occurs when X-rays are scattered by the regularly spaced atoms in a crystal lattice. The law provides a mathematical relationship between the angles at which X-rays strike the crystal and the distances between atomic planes within the crystal. When the angles match those predicted by Bragg’s Law, constructive interference occurs, resulting in observable diffraction patterns that hold valuable information about the crystal’s structure.
3. How does Bragg’s Law enable scientists to determine molecular structures?
Bragg’s Law allows scientists to decipher molecular structures by analyzing the diffraction patterns produced when X-rays pass through a crystal. When X-rays are incident at angles that satisfy the conditions of Bragg’s Law, they undergo constructive interference after interacting with the crystal lattice. This leads to distinct diffraction peaks in the resulting pattern. By measuring the angles and intensities of these peaks, researchers can mathematically deduce the arrangement of atoms within the crystal, providing insights into the molecule’s three-dimensional structure.
4. What are the practical applications of Bragg’s Law?
Bragg’s Law has numerous practical applications across scientific disciplines. In chemistry, it has been used to determine the structures of complex molecules, aiding in drug discovery and the development of new materials. In materials science, Bragg’s Law helps researchers understand the arrangement of atoms in various materials, contributing to the design of advanced materials with specific properties. Additionally, it plays a crucial role in fields like physics, biology, and geology, where understanding crystal structures is essential for unlocking the secrets of the natural world.