Valency is a fundamental concept in chemistry that plays a crucial role in understanding how elements interact and form compounds. It refers to the combined capacity of an atom or the number of electrons an atom can gain, lose, or share when forming chemical bonds with other atoms. Valency provides insights into the stability and reactivity of elements, enabling scientists to predict the types of compounds they can form and how they will interact with other substances.
At the heart of valency lies the concept of the electron configuration of an atom’s outermost energy level, also known as the valence shell. This outermost shell contains electrons that are most involved in chemical reactions. The main goal of atoms is to achieve a stable electron configuration, often resembling that of noble gases, which are highly stable due to their full valence shells.
Valency is determined by the number of valence electrons an atom possesses. The periodic table, a tool central to chemistry, provides valuable information about an element’s valency. Elements in the same column, or group, of the periodic table, often have similar valencies because they have the same number of valence electrons.
Here’s how valency works:
Valence Electrons: Elements in the same group of the periodic table share the same number of valence electrons. For example, all elements in Group 1 (such as hydrogen, lithium, and sodium) have one valence electron, and those in Group 17 (such as fluorine, chlorine, and bromine) have seven valence electrons.
Valence Shell Configuration: The valence electron configuration of an atom determines its valency. For instance, elements with one to four valence electrons typically lose those electrons to achieve a stable configuration with a complete valence shell. Elements with five to seven valence electrons tend to gain electrons to complete their valence shell. Elements with eight valence electrons, like noble gases, are already stable and rarely form chemical bonds.
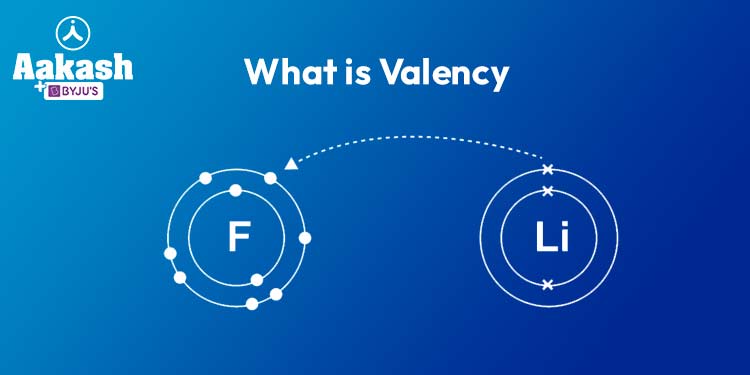
Chemical Bonds: Valency plays a crucial role in the formation of chemical bonds. Atoms interact to achieve a full valence shell by either gaining, losing, or sharing electrons. This results in the formation of different types of bonds, such as ionic bonds (electrons are transferred) and covalent bonds (electrons are shared).
Predicting Compound Formation: Understanding valency helps predict the types of compounds that elements can form. Elements with complementary valencies are more likely to combine and create stable compounds. For instance, sodium (valency 1) and chlorine (valency 1) readily form sodium chloride (table salt) due to their matching valencies.
Valency and Reactivity: Valency influences an element’s reactivity. Elements with similar valence electron configurations tend to have similar chemical properties and reactions. For example, Group 1 elements are highly reactive metals because they have one valence electron that they readily lose to achieve stability.
In summary, valency is a fundamental concept in chemistry that helps explain the interactions between atoms, the formation of compounds, and the behavior of elements. By understanding an element’s valency, scientists can predict its chemical behavior, which is essential for fields ranging from materials science to drug discovery. Valency underscores the elegance and predictability of the natural world at the atomic level.
Frequently Asked Questions (FAQs) About Valency in Chemistry
1. What is valency in chemistry?
Valency refers to the combined capacity of an atom, specifically the number of electrons it can gain, lose, or share when forming chemical bonds with other atoms. It plays a crucial role in determining how elements interact and combine to form compounds.
2. How is valency determined?
Valency is determined by the number of valence electrons an atom possesses, which are located in its outermost energy level or valence shell. The valence electron configuration dictates whether an atom will lose, gain, or share electrons to achieve a more stable electron configuration.
3. How does the periodic table relate to valency?
The periodic table provides valuable information about an element’s valency. Elements within the same group or column often share similar valencies because they have the same number of valence electrons. This arrangement allows chemists to predict how elements will react and form compounds.
4. What role does valency play in chemical bonding?
Valency is central to the formation of chemical bonds. Atoms interact to achieve a full valence shell and greater stability. Depending on their valencies, atoms can form different types of bonds, including ionic bonds (electron transfer) and covalent bonds (electron sharing).
5. How does valency influence compound formation and reactivity?
Valency is a key factor in predicting the types of compounds elements can form. Elements with complementary valencies are more likely to combine and create stable compounds. Valency also affects an element’s reactivity; elements with similar valence electron configurations tend to exhibit similar chemical properties and reactions.
6. Can valency help us understand the behavior of noble gases?
Yes, valency explains why noble gases are chemically inert. Noble gases have a full valence shell with eight electrons (except helium, which has two). This stable configuration makes them unlikely to form bonds with other elements, resulting in their minimal reactivity.
7. Why are elements in the same group likely to have similar valencies?
Elements in the same group of the periodic table share similar valencies because they have the same number of valence electrons. This commonality leads to comparable chemical behaviors and tendencies to form similar types of bonds and compounds.
8. How is valency relevant in practical applications?
Valency has significant implications in various fields, including materials science, pharmaceuticals, and environmental studies. Understanding valency helps scientists design and predict the behavior of molecules, reactions, and compounds, contributing to innovations in technology and medicine.
9. Do all elements follow the same valency patterns?
While certain trends exist based on an element’s position in the periodic table, there can be exceptions and variations due to factors like electron shielding and atomic structure. Transition metals, for instance, often exhibit multiple valencies depending on the specific compound they form.
10. Can valency change in different compounds?
Yes, an element’s valency can vary in different compounds, especially for transition metals. These elements can exhibit different valencies depending on the specific chemical environment and the type of compound they are forming. This versatility contributes to their ability to participate in a wide range of reactions and bond formations.