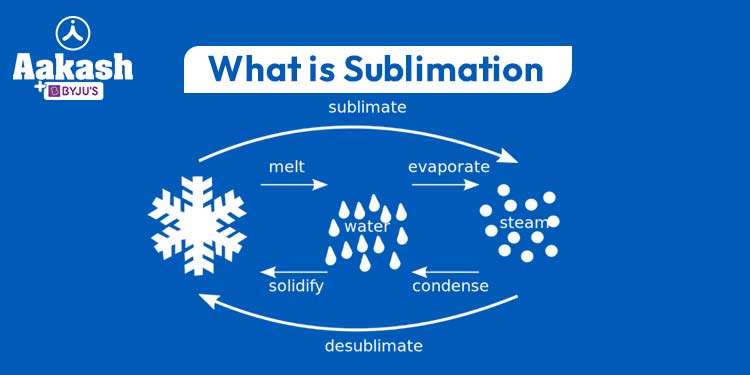
Sublimation is a fascinating phenomenon that challenges our understanding of how matter changes states. While most substances undergo changes in state between solid, liquid, and gas phases through processes like melting, freezing, evaporation, and condensation, sublimation presents a unique departure from this norm. It involves the direct transition of a substance from a solid to a gas, bypassing the liquid state entirely. This intriguing process has captured the curiosity of scientists and enthusiasts alike, leading to a deeper exploration of its principles and applications.
The Science Behind Sublimation:
At the heart of sublimation lies the interplay between temperature and pressure. Sublimation occurs when a substance’s vapor pressure at its solid state surpasses the surrounding atmospheric pressure, causing it to transition directly into a gas. Unlike melting or boiling, which require specific temperature changes, sublimation is governed by the substance’s unique vapor pressure characteristics. As the substance gains sufficient energy from its surroundings, its solid particles break free and enter the gas phase, forming a gaseous substance that retains the chemical composition of the original solid.
Common Examples of Sublimation:
Dry Ice (Solid Carbon Dioxide): One of the most well-known examples of sublimation is dry ice. At standard atmospheric pressure, solid carbon dioxide sublimates directly into carbon dioxide gas without becoming a liquid. This property makes dry ice a popular choice for creating eerie fog effects in theatrical productions and for cooling purposes in various industries.
Mothballs: Mothballs, often used to protect clothing from moth infestations, sublimate over time, releasing a gas that repels insects. This gradual transition from solid to gas ensures a continuous release of the repellent, making it an effective pest control method.
Snow and Frost: During cold winter nights, frost forms on surfaces as water vapor in the air sublimates directly into ice crystals. Similarly, snow can sublimate in extremely dry and cold conditions, turning directly into water vapor without melting first.
Applications of Sublimation:
Food Freeze-Drying: Sublimation plays a crucial role in freeze-drying, a preservation method widely used in the food industry. By freezing a food item and then subjecting it to reduced pressure, the ice within the food sublimates, leaving behind a dehydrated product that retains its original taste, texture, and nutritional value.
3D Printing: Sublimation is harnessed in 3D printing using a process called dye-sublimation printing. In this method, solid dye particles are transformed directly into a gas, which permeates a special printing paper and binds to a substrate, such as fabric or ceramics, producing vibrant and durable images.
Scientific Research: Sublimation is employed in various scientific studies, such as the analysis of volatile compounds and the purification of substances. It is also relevant in the field of astrophysics, where the sublimation of volatile molecules from comets and icy bodies contributes to the formation of their distinctive tails.
Conclusion:
Sublimation is a remarkable phenomenon that defies traditional expectations of matter’s state transitions. Its unique characteristics have paved the way for innovative applications across different industries, and its study continues to unravel mysteries in both the physical and chemical realms. As researchers delve deeper into the science of sublimation, we can expect to uncover even more remarkable insights and applications that harness this extraordinary process.
FAQs About Sublimation
1. What is sublimation?
Sublimation is a physical process in which a substance transitions directly from a solid state to a gas state without passing through the liquid phase. Unlike common state changes like melting or boiling, sublimation occurs when a substance’s vapor pressure at its solid state exceeds the surrounding atmospheric pressure.
2. How does sublimation work?
Sublimation occurs when a substance absorbs enough energy from its surroundings to break the bonds holding its solid particles together. As these particles gain energy, they transform into a gas and disperse into the surrounding environment, bypassing the liquid phase. This process is influenced by factors like temperature and pressure.
3. What are some examples of sublimation in everyday life?
Dry ice, often used for cooling and theatrical effects, is a well-known example of sublimation. Mothballs gradually release a gas through sublimation to repel insects, and frost forms on surfaces when water vapor sublimates into ice crystals. Sublimation is also involved in processes like freeze-drying foods and dye-sublimation printing.
4. How is sublimation used in industrial applications?
Sublimation finds application in various industries. In the food industry, freeze-drying uses sublimation to preserve the quality of food products. Dye-sublimation printing is utilized for creating high-quality images on fabrics, ceramics, and other materials. Additionally, sublimation is employed in scientific research, particularly in the analysis of volatile compounds and purification processes.
5. Can all substances undergo sublimation?
Not all substances can undergo sublimation. Sublimation requires specific conditions where the vapor pressure of the substance’s solid state exceeds the surrounding atmospheric pressure. Substances with lower vapor pressures at their solid states might not exhibit sublimation under normal circumstances. However, some substances may undergo sublimation under extreme conditions, such as very low temperatures and high vacuums.