The latent heat of fusion is the amount of energy required to change the physical state of any substance to its liquid state at a constant temperature. This concept was identified by a Scottish scientist named Joseph Black who recorded its value between 1750 and 1762. Latent heat of fusion is derived from the term “Latent heat”. Latent heat is the energy required to change the state of matter from solid to liquid, liquid to solid or liquid to gas. Keep scrolling through the article to learn more about “latent heat” and “latent heat of fusion”.
What is Latent Heat?
Latent heat is a fundamental concept in thermodynamics and plays a crucial role in understanding the behavior of substances during phase changes. The term “latent” means hidden, and latent heat refers to the heat energy absorbed or released by a substance during a change in its physical state—such as from solid to liquid or liquid to gas—without a change in temperature. This blog delves into the basics of latent heat, its types, and its significance in everyday phenomena.
Understanding Latent Heat
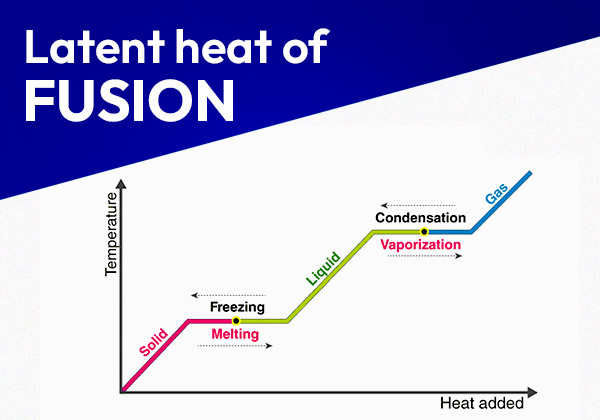
When a substance undergoes a phase change, such as melting, boiling, or freezing, the energy supplied or removed does not change the substance’s temperature but instead alters its internal energy. This energy, which is either absorbed or released during the phase transition, is known as latent heat.
For example, consider the process of boiling water. As heat is applied, the temperature of the water rises until it reaches 100°C (at standard atmospheric pressure). At this point, despite continuous heating, the temperature remains constant, and the water begins to convert into steam. The energy provided during this phase change is used to overcome the intermolecular forces holding the water molecules together in a liquid state, allowing them to move apart and form a gas. This energy is the latent heat of vaporization.
Types of Latent Heat
There are two primary types of latent heat:
Latent Heat of Fusion:
This is the heat energy required to change a substance from a solid to a liquid (melting) or from a liquid to a solid (freezing). For example, when ice melts to form water, it absorbs latent heat without any change in temperature.
Latent Heat of Vaporization:
This is the heat energy required to change a substance from a liquid to a gas (vaporization) or from a gas to a liquid (condensation). For instance, when water boils and turns into steam, it absorbs latent heat.
Both types of latent heat are essential for understanding the energy dynamics in various physical processes.
Significance of Latent Heat
Latent heat is a critical concept in numerous natural and industrial processes. Here are a few examples of its significance:
Climate and Weather Patterns:
Latent heat plays a significant role in the Earth’s climate system. The heat absorbed by water during evaporation from oceans, lakes, and rivers is released into the atmosphere when the water vapor condenses to form clouds, driving weather patterns.
Refrigeration and Air Conditioning:
The principles of latent heat are fundamental to the operation of refrigerators and air conditioners. These devices use the latent heat of vaporization of refrigerants to absorb heat from the surroundings, cooling the environment.
Phase Change Materials (PCMs):
Latent heat is harnessed in materials known as phase change materials, which store and release thermal energy during phase transitions. These materials are used in applications such as thermal energy storage and temperature regulation.
Who Discovered Latent Heat?
The concept of latent heat was first discovered by Joseph Black, a Scottish physicist and chemist, in the 18th century. Joseph Black conducted experiments in the 1750s and 1760s that led to the discovery of both latent heat and specific heat. His work laid the foundation for the study of thermodynamics and significantly advanced the understanding of heat and temperature.
Black’s experiments demonstrated that when a substance undergoes a phase change, such as melting or boiling, it absorbs or releases heat without a corresponding change in temperature. This observation led him to introduce the term “latent heat,” derived from the Latin word “latere,” meaning “to lie hidden.” His discovery was crucial in developing later theories of heat and energy.
The SI unit of Latent Heat of Fusion is J/kg.
Here’s a table representing Latent heat of Fusion for different substances in daily life:
| Elements | Latent Heat of Fusion (J/g) | Latent Heat of Fusion (Cal/g) |
| Aluminum (Al) | 397 | 94.89 |
| Chlorine (Cl2) | 181 | 43.26 |
| Copper (Cu) | 209 | 49.95 |
| Gold (Au) | 63.7 | 15.22 |
| Hydrogen (H2) | 59.5 | 14.22 |
| Iron (Fe) | 247 | 59.03 |
| Lead (Pb) | 23 | 5.5 |
| Mercury (Hg) | 11.4 | 2.72 |
| Nitrogen (N2) | 25.3 | 6.05 |
| Oxygen (O2) | 13.7 | 3.27 |
| Silver (Ag) | 105 | 25.1 |
| Sodium (Na) | 113 | 27.01 |
| Tungsten (W) | 285 | 68.12 |
| Zinc (Zn) | 112 | 26.77 |
Latent Heat of Fusion Formula
Here is the formula that is used in the world of thermodynamics to calculate the latent heat of fusion,
Q = mL + mcΔT
- Q is the latent heat of fusion
- M is the mass of body of substance
- L is the specific heat of fusion
- C is the specific heat of substance
- ΔT is the temperature that is changing during the absorption of heat.
Here are the values for specific heat of fusion of different elements:
- Specific Heat of Fusion of Water: 334 J/g or 79.7 cal/g
- Specific Heat of Fusion of Lead: 24.5 J/g or 5.85 cal/g
- Specific Heat of Fusion of Iron: 272 J/g or 65 cal/g
- Specific Heat of Fusion of Copper: 205 J/g or 49 cal/g
- Specific Heat of Fusion of Gold: 63 J/g or 15 cal/g
Daily Life Implications of Latent Heat of Fusion
Here are some daily life activities that uses the concept of latent heat of fusion for better understanding:
Example 1 Cooling drinks
To fasten the process of cooling drinks, you add cold water or ice. When the ice melts, a large quantity of heat is absorbed from the drinks and it makes the drink come to a temperature that is the melting limit of ice.
Example 2 Melting the ice on road by salt
The specific latent heat of fusion of salt is higher than ice. When you put the salt on a road which has a thick block of ice, the salt that requires more energy absorbs it from the ice. This results in melting of the ice.
Latent Heat of Fusion FAQs
Q1. What is latent heat?
Answer: Latent heat is the energy that is either released or absorbed by the body or thermodynamic system during a constant-temperature process. The two major forms of latent heat are melting (latent heat of fusion), and boiling (latent heat of vaporization).
Q2. What is the SI unit of latent heat?
Answer: The SI unit of latent heat is J/kg.
Q3. What is the latent heat of fusion?
Answer: Latent heat of fusion is the amount of heat required to or absorbed when a solid changes to liquid at its melting point in atmospheric pres is known as latent heat of fusion.
Q4. What are the other names for latent heat?
Answer: The other terms used to describe latent heat are latent energy and heat of transformation.
Q5. Who introduced the concept of latent heat?
Answer: The concept of latent heat was first introduced by Joseph Black between 1750 and 1762. He was a scottish scientist who noted down the latent heat values. Later, a British physicist named Prescott Joule described the latent heat as energy.