The characteristics of particles of matter are fundamental to understanding the nature of substances around us. Matter is composed of tiny particles, such as atoms, molecules, and ions, that exhibit specific properties and behaviors. These particles are the building blocks of all materials and dictate how substances interact, transform, and function.
Understanding these characteristics is essential for grasping basic concepts in chemistry and physics. The arrangement, type, and interactions of particles influence the physical and chemical properties of matter, such as its state, density, and reactivity. By studying particles, scientists can predict and control material behavior, design new materials, and develop technologies in various fields.
In essence, the study of particle characteristics provides insights into the structure and dynamics of matter, driving advancements in science and industry and enhancing our ability to understand and manipulate the world at a microscopic level.
Types of Particles of Matter
- Atoms: The smallest units of matter that retain chemical properties. Atoms consist of a nucleus (containing protons and neutrons) and electrons orbiting around the nucleus.
- Molecules: Combinations of two or more atoms bonded together. Molecules can be simple, like oxygen (O₂), or complex, like DNA.
- Ions: Charged particles formed when atoms or molecules gain or lose electrons. Ions can be positive (cations) or negative (anions).
- Subatomic Particles: Fundamental particles that make up atoms, including protons, neutrons, and electrons.
Functions of Particles of Matter
Formation of Elements and Compounds: Atoms combine to form elements, and different elements combine to form compounds, which are essential for the chemical processes that sustain life.
- Chemical Reactions: Particles interact and rearrange during chemical reactions, leading to the formation of new substances.
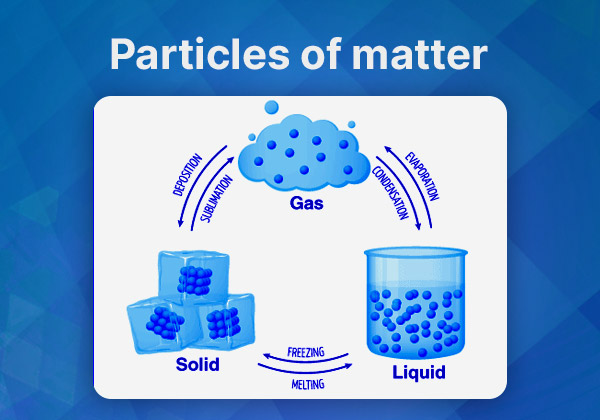
- Physical Properties: The arrangement and type of particles determine the physical properties of matter, such as state (solid, liquid, gas), density, and melting/boiling points.
Importance of Particles of Matter
- Fundamental Building Blocks: Particles are the building blocks of all matter, making them crucial for understanding the structure and behavior of materials.
- Basis for Chemical Reactions: Understanding particle interactions helps predict and control chemical reactions, which is vital for industries, medicine, and environmental science.
- Explaining Physical Phenomena: The study of particles helps explain various physical phenomena, including phase changes, diffusion, and the properties of materials.
Uses of Understanding Particles of Matter
- Material Science: Knowledge of particle characteristics aids in developing new materials with desired properties, such as stronger metals or more efficient semiconductors.
- Medicine: Understanding how particles interact at the molecular level contributes to drug design and the development of new treatments.
- Environmental Science: Insights into particle behavior help in pollution control and developing methods to reduce environmental impact.
Effects of Particle Behavior
- States of Matter: Particle arrangements affect whether a substance is a solid, liquid, or gas.
- Chemical Stability: Particle interactions determine the stability and reactivity of substances, influencing their safety and usability.
- Biological Functions: Particle behavior is crucial for biological processes, including enzyme function and cellular processes.
In summary, the characteristics of particles of matter are central to the science of chemistry and physics. They help explain the nature of materials, how they interact, and their practical applications in various fields. Understanding these properties not only provides insight into the fundamental nature of matter but also drives advancements in technology, medicine, and environmental science.
Characteristics of Particles of Matter FAQs
Q1: What are the primary types of particles of matter?
Answer: The primary types of particles of matter include atoms, molecules, ions, and subatomic particles. Atoms are the smallest units of matter, molecules are combinations of atoms, ions are charged particles, and subatomic particles are the fundamental components of atoms, such as protons, neutrons, and electrons.
Q2: How do particles of matter function in chemical reactions?
Answer: In chemical reactions, particles interact and rearrange to form new substances. Atoms in molecules break and form new bonds, leading to the creation of different compounds. This process is essential for the formation of new materials and for chemical transformations in various industries.
Q3: Why is understanding particles of matter important?
Answer: Understanding particles of matter is crucial because it helps explain the structure and behavior of materials, predict and control chemical reactions, and understand physical phenomena. This knowledge is foundational in fields such as material science, medicine, and environmental science.
Q4: How does the behavior of particles affect the physical properties of matter?
Answer: The arrangement and type of particles determine the physical properties of matter, such as its state (solid, liquid, or gas), density, and melting/boiling points. For example, the close packing of particles in a solid results in its rigidity, while the loose arrangement in a gas allows it to expand and fill its container.
Q5: What are some practical uses of understanding particle characteristics?
Answer: Understanding particle characteristics is applied in various fields such as:
Material Science: For developing new materials with specific properties.
Medicine: In drug design and treatment development.
Environmental Science: To address pollution and reduce environmental impact.