Preparation of Lyophobic Colloid - Condensation or Aggregation Methods, Dispersion Methods, Practice Problems and FAQs
Are you familiar with the color of pure gold?
The majority of you said yellow, right?
But in reality, The size of the particles is a factor. Red is the color of the purest gold. The particle changes color as it becomes bigger, going from purple to blue to golden yellow.
Now, how can we prepare gold that is coloured red?
This lyophobic gold sol has particles that range in size from 1 to 1000 nm. in order to comprehend how this gold sol was made.
First, let's talk about how to prepare lyophobic sols!
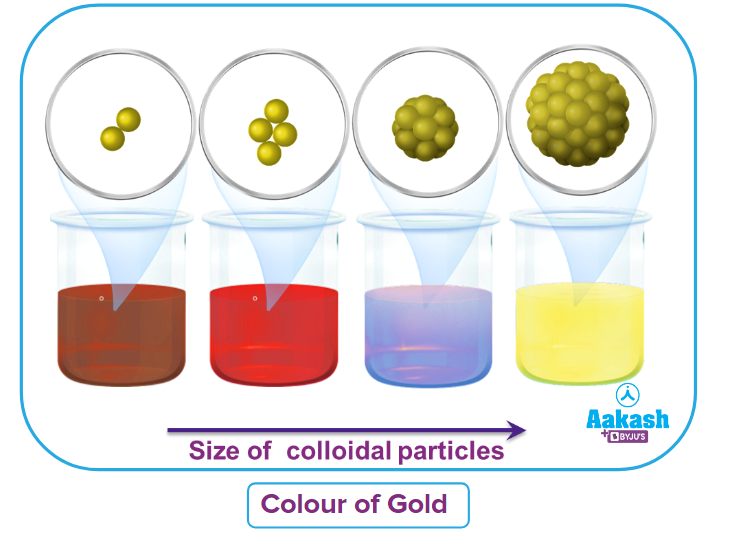
Table of Contents:
- Lyophobic sols
- Preparation of Lyophobic colloids
- Condensation or Aggregation methods
- Dispersion Methods
- Practice Problems
- Frequently Asked Questions-FAQs
Lyophobic sols:
Lyophobic sols are those that cannot be produced by mixing the dispersed phase with the dispersion medium. Metals and their sulphides are two examples. Their colloidal solutions can be made using certain techniques, and these sols are easily precipitated when electrolytes are added in modest amounts.
Preparation of Lyophobic colloids:
Lyophobic sols are prepared using two major sorts of techniques:
1) Condensation or Aggregation methods
2) Dispersion methods
Condensation or Aggregation methods:
These techniques condense smaller dispersed phase particles into colloidal-sized particles. These techniques are used to do this:
a) Through chemical reactions:
Lyophobic colloidal solutions can be made using the chemical processes listed below.
(i) Oxidation: By bubbling H2S gas through Br2 water, sulphur dioxide, etc., a colloidal sol of sulphur is produced.
(ii) Reduction: By reducing the compounds of metals, colloidal solutions of the metals are produced.
As an illustration, SnCl2 is used to decrease a solution of AuCl3.
Note: The purple-coloured gold sol created in this manner is referred to as Cassius's purple.
Formaldehyde can also be used to carry out the reaction.
(iii) Hydrolysis: When a solution of concentrated ferric chloride is introduced drop by drop to hot water, a colloidal solution of ferric hydroxide is created.
(iv) Double decomposition: Arsenious oxide in diluted solution in water is subjected to H2S to produce As2S3 sol.
(b) Through Excessive Cooling:
By freezing a water solution in the solvent, colloidal solution of ice in an organic solvent like ether or chloroform can be formed. Water molecules that cannot be retained for a long time in solution mix on their own to create colloidal-sized particles.
(c) By Solvent Exchange:
By adding more water than necessary to the alcoholic solution of some compounds, like phosphorus and sulfur, which are insoluble in water but soluble in alcohol, the colloidal solution can be created.
(d) By a change in the physical state:
Sols of elements such as mercury and sulfur are created by passing their vapors through cold water that has been stabilized with an appropriate agent, such as citrate or ammonium salt.
Dispersion Methods:
These techniques include breaking down a substance (suspension) into smaller particles.
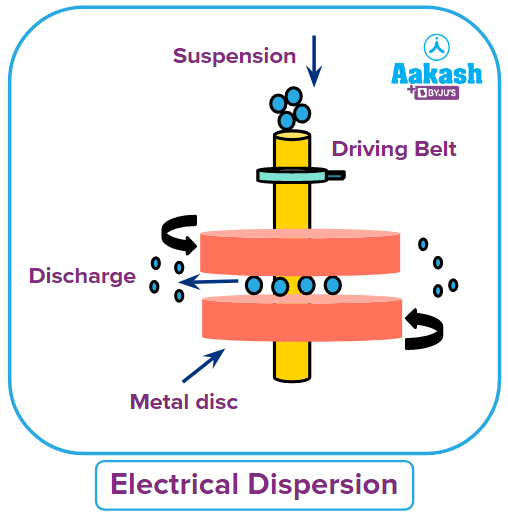
(a) Mechanical dispersion:
This method is used to initially grind the material to gritty particles. The dispersion medium is then combined with it to create a suspension. A colloidal mill is then used to grind the suspension. It is made up of two fast-spinning metallic discs that are almost touching and revolving in opposing directions.
The mill's disc spacing has been tuned in such a way that the coarse suspension undergoes intense shearing, producing particles with a colloidal size. This technique can be used to create colloidal solutions of black ink, paint, varnish, dyes, etc.
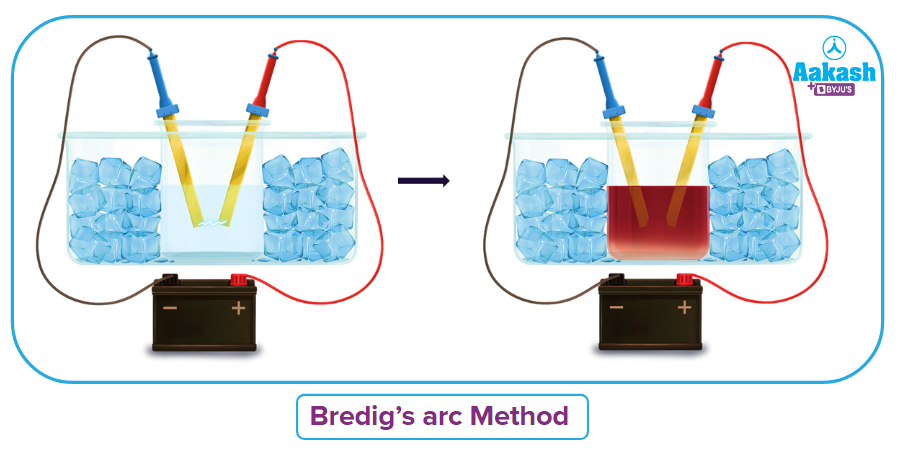
(b) Using the Bredig's arc method:
Sols of metals such as Pt, Ag, Au and Cu are created using this technique.
Two electrodes submerged in a dispersion media, such as water, are used to create the metal whose sol is to be created. By enclosing the dispersion medium with a freezing mixture, the temperature is maintained. Between the electrodes is created an electric arc. The metals are vaporized by the arc's intense heat and then instantly condense in the liquid to form a colloidal solution. The prepared colloidal solution is stabilized by a little addition of KOH to it.
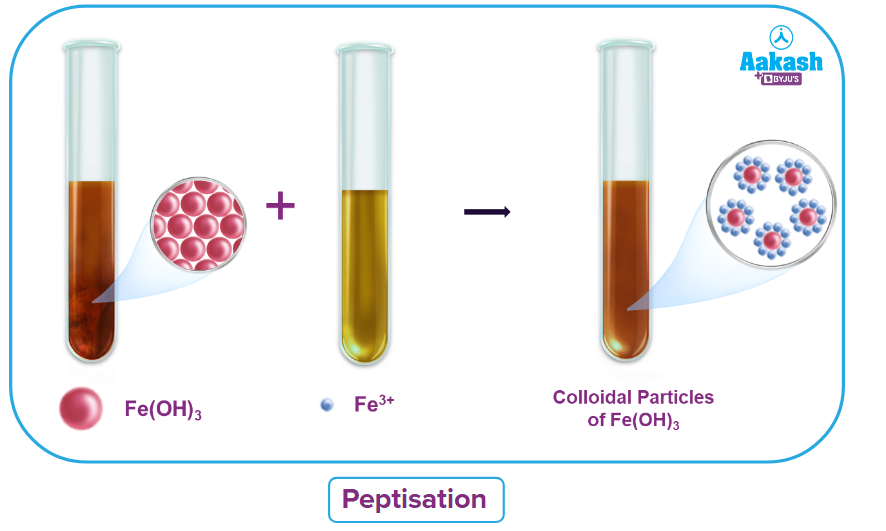
(c) Through Peptization:
By adding the proper electrolyte, peptization is the process of converting a freshly prepared precipitate into colloidal solution.
Peptizing agents are the electrolytes employed in this process.
What Causes Peptization?
The appropriate ions from the additional electrolyte are absorbed by the precipitate particles when an electrolyte is added to a freshly prepared precipitate. A colloidal solution is created when the charged particles repel one another.
As an illustration, a small amount of FeCl3 solution added to a precipitate of iron (III) hydroxide results in the formation of a colloidal solution that is reddish brown in colour.
Fe(OH)3 with Fe3+ results in Fe(OH)3Fe3+.
A colloidal solution of silver chloride can be created by peptising a precipitate of silver chloride with a diluted solution of silver nitrate.
AgCl with Ag+ results in the formation of a colloidal solution of AgClAg+.
Practice Problems:
Q1. Which of the following is the true statement for Peptization?
a. By adding a lyophilic sol, a freshly generated precipitate is converted into a colloidal form during the process of peptization.
b. The electrolytes used in this procedure are coagulating agents.
c. When an electrolyte is added to a freshly produced precipitate, the particles present in the precipitate take up the necessary ions from the electrolyte and colloidal sol prepared as a result of repulsion of the same charge between particles and particles of electrolyte.
d. All of these truly explain Peptization
Answer: (C)
Solution: In peptization, colloidal sol is prepared by mixing a freshly generated precipitate in an electrolyte, not in a lyophilic sol. So, option (A) is an inappropriate statement.
The electrolytes used in peptization to prepare colloidal sols are peptizing agents not coagulating agents. Hence, option (B) is also inappropriate.
Option (C) is correct as it is true that When an electrolyte is given to a freshly formed precipitate, the precipitate particles absorb the required ions from the electrolyte and colloidal sol created as a result of the repulsion of the same charge between the precipitate particles and the electrolyte's particles.
Hence, the correct option is (C).
Q2. Which of the following methods falls under the category of condensation methods used for the preparation of lyophobic sols?
a. Bredig's arc method
b. Peptization
c. Both of these
d. None of these
Answer: (D)
Solution: Condensation or aggregation procedures and dispersion methods are the two main types of techniques that can be used to create lyophobic sols. The preparation procedures for the condensation process include chemical reactions, excessive cooling, solvent exchange, a change in the physical state, and more. Electric dispersion, peptization, and Bredig's arc method are the three procedures that make up the dispersion method. Hence, both methods that are given belong to the dispersion method, not to the condensation method.
Hence the correct answer is option (D).
Q3. Which of the following given statements is suitable for the Electrical dispersion method?
a. This method is used to initially grind the suspension into colloidal particles.
b. Two swiftly rotating metallic discs that are virtually touching and rotating in opposition to one another make up this device.
c. The coarse suspension experiences strong shearing and produces particles with a colloidal size thanks to the mill's finely calibrated disc spacing.
d. All of these
Answer: (D)
Solution: Electrical dispersion is a method which is used to prepare lyophobic sol by using a dispersion technique in which large-sized particles like the suspension are broken into the size of the colloidal particle using the colloidal mill. So, option (A) is correct.
The colloidal mill contains two metallic discs which are very close and rotating in opposite directions as shown in the image. Hence option (B) is also correct.
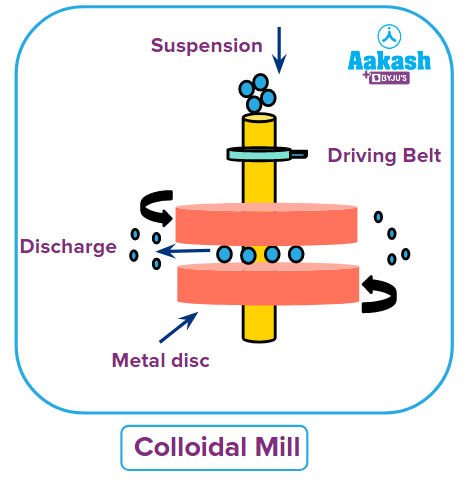
The mill's highly adjusted disc spacing results in vigorous shearing or tearing of the suspension and creates particles with a colloidal size. Hence this option (C) is also correct.
Hence the correct answer is an option (D).
Q4. Cassius's purple is prepared by which of the following given methods?
a. By reduction using dispersion method
b. By reduction using condensation method
c. By Bredig’s Arc method using condensation method
d. By Bredig’s Arc method using dispersion method
Answer: (B)
Solution: A purple pigment known as "purple of Cassius" is created when gold salts and tin(II) chloride combine.
The purple-coloured gold sol created in this manner is referred to as Cassius's purple. Here gold reduced from Au3+to Au. Hence this is a reduction method which falls under the category of condensation method or aggregation method.
Hence the correct answer is option (B).
Q5. Which stabilizer is used in the preparation of mercury sols?
a. Citrate
b. Ammonia
c. Sulfides
d. All of these
Answer: (A)
Solution: Ammonium salts and citrate are used as stabilizers in preparation of mercury sols using methods of change in physical state which comes under the category of condensation.
Hence the correct answer is an option (A)
Frequently Asked Questions-FAQs:
1. Why do lyophobic sols coagulate so easily?
Answer: Since only charge determines the stability of lyophobic sols, they are less stable. On the other hand, Lyophilic sols are more stable because the particles' charge and solvation contribute to their stability. Lyophobic sols can thus be coagulated with ease.
2. How may a sol who is lyophobic be protected?
Answer: The inclusion of lyophilic sol can protect lyophobic sol. Lyophilic sol is added to lyophobic sols to stabilize them because lyophobic sols rapidly precipitate with the addition of modest amounts of electrolytes, shaking, or heating.
3. Are hydrophobic and lyophobic the same thing?
Answer: They are referred to as hydrophilic when using water as the dispersion medium. Lyophobic sols or colloids are solvent-hating. They are referred to as hydrophobic when water is the dispersion medium.
4. Is blood a Lyophobic colloid?
Answer: Solid cells suspended in liquid blood plasma makeup colloidal blood. Blood is a lyophilic colloid because cells and plasma can be separated and mixed together in hospitals. Hence, it is not a lyophobic colloid.
5. What distinguishes solutions from colloids, exactly?
Answer: The solute particles in a solution are frequently smaller than the particles in a colloid. When compared to colloids, which can also be a heterogeneous mixture, solutions are fully homogeneous. Solutions are transparent, whereas colloidal mixes look to be opaque or translucent.

