-
Call Now
1800-102-2727
Structure of ketone: Structure, physical properties and uses of ketones, practice problems & Frequently asked questions(FAQs)
When looking at the furniture in the house, the wood may appear to be covered with some protective coating.
A varnish is a type of protective film.

Wood varnish is an almost necessary component of any woodwork.
This is a popular method characterised by the application of a resinous substance with a protective function that imparts a glossy appearance to the furniture. The paint, on the other hand, deteriorates over time and seems corroded and dirty. For a long time, this problem did not exist in everyday furniture, but it can spoil the beauty of oil paint masterpieces. Acetone is a widely used chemical for removing bad paint. Rub a soft, fine steel wool pad dampened with acetone on the wood of furniture.
Table of content:
- Structure of ketone
- Physical properties of ketone
- Uses of ketone
- Practice problems
- Frequently asked questions(FAQs)
Structure of ketone:
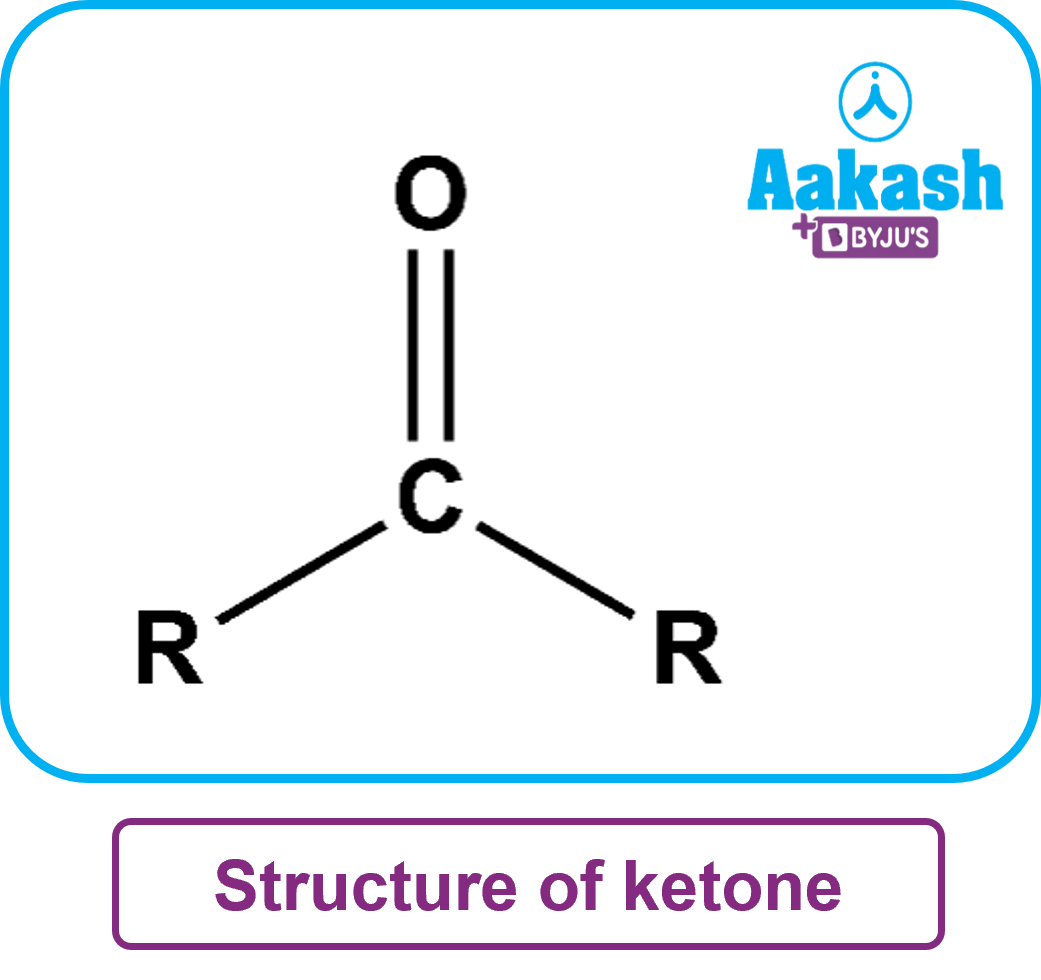
Ketones are organic compounds that have the functional group C=O and the structural formula R (C=O) R'. R and R' can be alkyl or aryl groups in this case. Carbon-containing substituents are present on both sides of the carbon-oxygen double bond in these carbonyl compounds. The ketone group's carbonyl carbon has an sp2 hybrid orbital. The ketone has a triangular planar structure. This structure's bond angle is around 120o. Ketones are nucleophilic at the oxygen atom and electrophilic at the carbon atom because carbon-oxygen interactions polarise carbonyl group(oxygen is more electronegative than carbon).
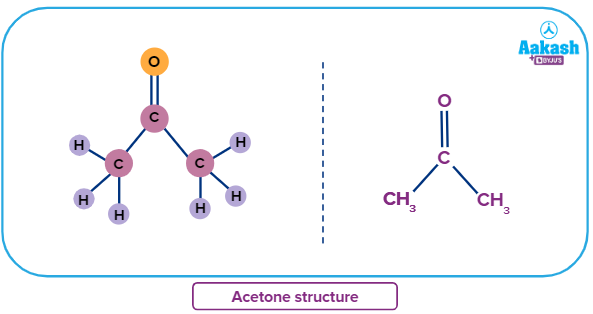
Now we will discuss one common ketone known as acetone.
Acetone (propanone) is an organic compound with the chemical formula (CH3)2CO. When using nail polish remover, a colourless, unpleasant stench may appear. Because of its composition and molecular structure, acetone is extremely important in chemistry. The acetone oxygen atom's lone pairs can partially attract hydrogen atoms and form hydrogen bonds. Because of this feature, acetone is the favoured solvent in the industry. In chromatography, for example, acetone is preferred over water because it can dissolve both polar and nonpolar dyes during analysis.
It is vital to note that acetone molecules do not dissociate into ions when dissolved in water. They are, however, totally blended to make a non-uniform combination. The acetone molecule's two methyl groups are nonpolar and dissolve nonpolar compounds, but the carbonyl group in the centre is polar and dissolves polar compounds.
Another feature that contributes to acetone's suitability as a solvent is its miscibility with other polar molecules, particularly water and other organic compounds. This enables you to create many types of industrial, home, and experimental solvents, depending on the use. Acetone is a non-toxic, organic chemical that is powerful and easily miscible with other substances. Acetone is a critical ingredient in the chemical industry, although various household items contain acetone.
Physical Properties of ketone:
Boiling point: The boiling points of ketones often increase with increasing molecular weight. The strength of the intramolecular force determines the boiling point.
- Van der Waals Dispersion Forces: As the length and number of atoms of a molecule increase, so do the forces of attraction between them. The boiling point of ketones increases as the number of carbon atoms increases.
- Van der Waals Dipole Dipole attractions: Ketones are inherently polar due to the existence of carbon-oxygen double bonds. A permanent dipole and a nearby molecule are attracted to each other. This is why ketones have greater boiling points than similar-sized hydrocarbons.
Solubility:
In water, ketones (up to four carbon atoms) are miscible. Although ketones cannot make hydrogen bonds with one another, they can form hydrogen bonds with water molecules. A hydrogen bond is formed when one of the slightly positive hydrogen atoms in a water molecule attracts one of the lone pairs on the oxygen atom of a ketone.
The presence of a hydrogen bond link between the polar carbonyl group and water molecules is responsible for ketone solubility in water.
However, the solubility of ketones in water diminishes significantly as the length of the alkyl chain (carbon chain) rises. The greasy alkyl chain begins to interfere with water solubility. They break the very strong hydrogen bonds between water molecules without replacing them with anything, making the process less profitable in terms of energy, and therefore decreasing solubility.
As a result, the higher members (those with more than four carbon atoms) are insoluble in water. Ketones, on the other hand, are soluble in organic solvents (like dissolves like), such as benzene, ether, chloroform, and alcohol.
Bonding:
The carbonyl (>C=O) functional group is made up of a carbon atom that is double-bonded to an oxygen atom. Oxygen is more electronegative than carbon, and as a result of this difference in electronegativity, oxygen has a strong tendency to pull electrons in a carbon-oxygen bond towards itself. This causes the formation of dipoles with a slight positive charge on the carbon atom and a slight negative charge on the oxygen atom. As a result, the carbon-oxygen double bond becomes extremely polar.
A nucleophile can target the slightly positive carbon atom in the carbonyl group, while electrophiles attack the slightly negative oxygen atom.
Uses of ketone:
- Ketone is an excellent solvent for certain polymers and synthetic fibres.
- Acetone can be used as a paint thinner and remove nail polish.
- It is also used for medical purposes, like as chemical peeling and acne treatments.
- Butanone, commonly known as methyl ethyl ketone, is a common solvent. It is used in the manufacture of textiles, varnishes, paint removers, paraffin wax, and plastics, etc.
- Another major ketone is cyclohexanone, which is used in the production of nylon.
- Propanone (acetone) is used in the production of polymers such as plexiglass.
- Ketones are used as a solvent and as a starting material for the synthesis of many organic compounds.
- Acetone and ethyl methyl ketone are mainly used as industrial solvents.
Practice problems:
Q.1. Ketones undergoes
(A) Nucleophilic addition reactions
(B) Electrophilic substitution reactions
(C) Nucleophilic substitution reactions
(D) Electrophilic addition reactions
Answer: (A)
Solution: Ketones contain a polar -C=O group with a double bond. The entering nucleophile attacks the sp2 hybridised carbon, breaking the double bond and converting it to sp3 resulting in the formation of a tetrahedral alkoxide intermediate. This goes through another fast step to form an addition product.
Q.2. Among the following which is least reactive in nucleophilic addition reaction?
(A) CH3(CO) CH3
(B) C6H5 (CO) CH3
(C) C6H5 (CO) C6H5
(D) All of these
Answer: (C)
Solution: The presence of relatively large substituents in ketones makes nucleophile approaches to carbonyl carbon difficult due to steric hindrance. Because the two alkyl groups in acetone are small, the nucleophilic addition reaction is easier in acetone than in the other two compounds and in benzophenone due to presence of two bulky groups and the electrophilicity of the carbonyl group in the other two compounds is reduced due to the presence of the phenyl ring.
Q.3. What is the common name of Butan-2- one?
(A) Acetone
(B) Ethyl Methyl Ketone
(C) Diethyl ketone
(D) Isopropyl methyl ketone
Answer: (B)
Solution: Ketones are generally named by writing the name of the individual alkyl groups attached to the carbonyl carbon, then by writing "ketone" as the third word in the name. For example, butan-2-one can be written as ethyl methyl ketone.
Q.4. Which of the following has a higher boiling point than ketones?
(A) Alcohol
(B) Alkanes
(C) Aldehyde
(D) Ether
Answer: (A)
Solution: Because of the presence of hydroxyl groups, alcohols are polar molecules with adequate intermolecular interaction. Because of the huge difference in electronegativity between oxygen and hydrogen atoms, the strength of dipole-dipole interaction is stronger in alcohol.
Alcohol molecules are capable of intermolecular hydrogen bonding and exist as associated molecules due to their high dipole-dipole interaction.
Because breaking the hydrogen bond created in the molecular network needs a substantial amount of energy, the boiling point of the alcohol molecule is high.
Because of stronger hydrogen bonding, alcohol has a higher boiling point than ketones.
Frequently asked questions:
Q1. Why are aldehydes more reactive than ketones in nucleophilic addition reactions?
Answer: In nucleophilic addition reactions, aldehydes are more reactive than ketones due to steric and electronic reasons. The presence of two relatively large substituents in ketones makes approaching the carbonyl carbon more difficult than in aldehydes with only one such substituent.Two alkyl groups in ketones lower the electrophilicity of the carbonyl carbon more efficiently than in aldehydes. As a result, Aldehydes are more reactive than Ketones in nucleophilic addition reactions.
Q2. Why is ketone oxidation more difficult than aldehyde oxidation?
Answer: You'll remember that the presence of a hydrogen atom connected to the carbon-oxygen double bond in the aldehyde distinguishes it from a ketone. Ketones lack this hydrogen. Because of the existence of that hydrogen atom, aldehydes are very easy to oxidise (i.e.they are strong reducing agents) and Ketones are resistant to oxidation because they lack a hydrogen atom linked to their carbonyl.
Q3. What is ketosis?
Answer: Ketosis is a metabolic condition in which your blood contains high levels of ketones, namely beta-hydroxybutyrate.
It happens when your body begins to use fat as its primary fuel source due to a lack of glucose, or blood sugar, which is usually caused by starvation, fasting, or following an extremely low carb diet. Many cells in the body prefer to run on glucose. When your body does not have enough glucose to power these cells, insulin levels drop, causing large amounts of fatty acids to be released from body fat storage.
Many of these fatty acids are transferred to the liver and oxidised, resulting in ketones, also known as ketone bodies. These are then used as an alternative source of energy throughout the body.
Q4. What causes acetone to be more volatile than alcohol?
Answer: Acetone, as a ketone, lacks hydrogen bonding due to the lack of direct -OH bonds. While ethanol, as an alcohol, has a direct -OH interaction. As a result, ethanol has intermolecular hydrogen bonding. As a result, more physical bonds must be broken in ethanol than in acetone. Hence acetone evaporates faster than ethanol.


