-
Call Now
1800-102-2727
Properties of Water-Introduction, Physical properties, Chemical properties, Structure of water, Uses, Practice Problem, FAQs
What would life be like without water?
What do you think about this? We can’t imagine such a drastic situation.
Water is a necessity for all living things to live. Without water, there is no life on Earth.
Have you asked yourself why water is so important? Mainly because water makes up about 60% of our bodily weight. In order to maintain other internal functions and to assist regulate body temperature, our bodies require water in all of the cells, organs, and tissues. We consume water so frequently that even someday we can survive without food, but it is very difficult for a living creature to survive without water. Drinking fluids and consuming meals that contain water are essential for rehydrating since our bodies lose water through breathing, sweating, and digestion.


Water is a very light gas when it is in its gaseous condition, but when it is liquid, it is even denser than its solid form. It is crucial for people to learn more about water because of its peculiar qualities, which have a significant impact on our daily lives.
To continue enjoying the benefits of clean water and healthy oceans and rivers, we need to fundamentally change how we use and treat water. Let us study some important properties of water in detail.
Table of content:
- Water
- Physical Properties of Water
- Chemical Properties of Water
- Structure of Water
- Uses of Water
- Practice Problems
- Frequently Asked Questions-FAQs
Water:
One of the most accessible and plentiful compounds is water. Water covers almost three-fourth of the earth's surface. It is necessary for life. Water makes up two thirds of the human body. Because it can dissolve so many different chemicals, it is especially significant to chemists. Water is used as a medium for a wide range of chemical processes as a result. Water occurs as a solid below 0°C and as a gas above 100°C, despite the fact that under normal atmospheric pressure it is most frequently regarded as a liquid. The solid and gaseous states of water are referred to as ice and steam, respectively.
Physical Properties of Water:
- The freezing point of water is 0℃ or 273.15 K at STP conditions.
- The boiling point of water is 100℃ or 373.15 K at STP conditions.
- The heat of vapourisation of water is 540 cal g-1 at 100℃ at STP conditions.
- The heat of fusion of water is 79.7 cal g-1 at 0℃ at STP conditions.
- The specific heat of water is 4184 J kg-1 K-1at STP conditions.
- The dipole moment of water is approx. 1.8546 D.
- The specific heat, thermal conductivity, surface tension, dipole moment, and dielectric constant of water are all higher than those of air.
- Since the density of ice is less than that of water, the ice floats over water.
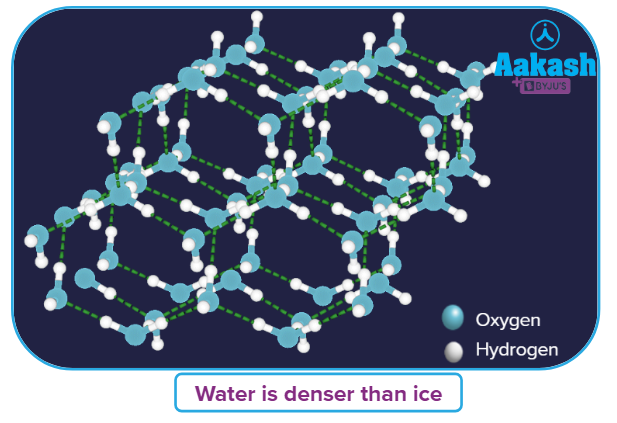
- Triple point (solid, liquid, and vapour all the three phases exit together): At 273.15 K, 0.006 atm, water is in equilibrium with ice and vapour.
- Pure water has no flavour, colour, or odour.
- It imparts a bluish tint in substantial layers.
- It has a propensity to link up. It exists as linked molecules through hydrogen bonding rather than as a single H2O molecule in the liquid state.
Chemical Properties of Water:
- The nature of water is neutral. Pure water has a pH of 7. It is a weak electrolyte that ionises into H+ and OH- ions sporadically.
H2O ⇌ H++OH-
In pure water, [H+]=[OH-]=10-7 at 25℃
- Reactions with Metals: It produces hydrogen gas when it combines with active metals. In the case of alkali and alkaline earth metals, the reaction is exothermic.
When steam is passed over heated metals, such as Zn, Mg, and Fe, it is broken down.
Only heat causes Pb and Cu to breakdown water. Metals such as Ag, Au, Hg, and Pt do not break down water.
- Reaction with nonmetals:
Fluorine decomposes or breaks cold water.
Chlorine decomposes cold water forming HCl and HClO.
However, when sunshine is present, only HCl is generated with the release of oxygen.
Water gas is formed when steam is applied to 1,000°C red-hot coke.
- Action on non-metallic oxides:
Acids are formed when acidic oxides and water interact.
CO2(g)+H2O(aq) ⇌ H2CO3(aq) (Carbonic acid)
SO2(g)+H2O(aq)H2SO3(aq) (Sulphurous acid)
SO3(g)+H2O(aq)H2SO4(aq) (Sulphuric acid)
P2O5(s)+3H2O(aq)2H3PO4(aq) (Orthophosphoric acid)
N2O5(s)+H2O(aq)2HNO3(aq) (Nitric Acid)
- Amphoteric nature
It is the ability of a substance to act as an acid as well as a base.
Example:
H2O(l)+NH3(aq) ⇌ OH-(aq)+NH4+(aq)
Here, water acts as a bronsted acid.
H2O(l)+H2S(aq) ⇌ SH-(aq)+H3O+(aq)
Here, water acts as a bronsted base.
Water undergoes auto-protolysis (self-ionization) as follows:
H2O(aq)+H2O(aq) ⇌ H3O+(aq)+ OH-(aq)
Amphoteric oxides or hydroxides are formed by several metals, including zinc, tin, lead, aluminium, and beryllium.
- Oxidising and reducing nature:
Highly electropositive metals can easily reduce water to evolve hydrogen gas.
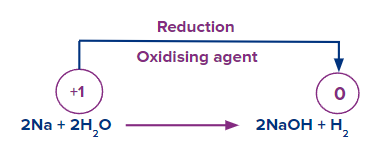
![]()
Water can be oxidised to dioxygen in the presence of fluorine.
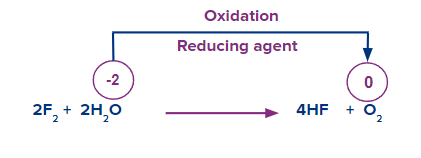
![]()
- Formation of hydrates with metals:
With metals, water creates three different types of hydrates. Many salts can crystallize as hydrated salts from aqueous solutions. There are various forms of water in such an arrangement.
Coordinated hydrates: In some compounds, water molecules are attached to the metal
ions through coordinate bonds (complex compounds). These types of hydrates are called
coordinate hydrates.
Example: [Fe(H2O)6]Cl3
Hydrogen bonded hydrates: In some compounds, water molecules are associated with
hydrogen bonds. These types of hydrates are called hydrogen-bonded hydrates.
Example: CuSO4.5H2O.
Interstitial hydrates: In some compounds, water molecules occupy the interstitial sites in
the crystal lattice. These types of hydrates are called interstitial hydrates.Example: BaCl2.2H2O.
- Water as a catalyst: In numerous processes, water serves as a catalyst. In general, perfectly dry gasses do not react, but moisture causes the chemical change. Only in the presence of moisture do gasses of ammonia and hydrochloric acid mix.
Structure of Water:
Water is a simple molecule. Its molecular structure is quite simple. It is made up of two hydrogen atoms and one oxygen atom. A shared pair of electrons bind each hydrogen atom to oxygen covalently. Oxygen also has two electron pairs that are not shared forming a bond angle of 104.5° between H-O-H bond. Each O-H bond is polar.
E.N. of O (3.5) > E.N. of H (2.1) (Both values are as per the Pauling Scale)
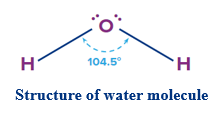
![]()
Uses of water:
Water can be used in a variety of ways, both for direct and indirect purposes.
- Direct uses of water are bathing, drinking, cooking, planting etc.
- Indirect uses include the processing of wood to manufacture paper and the production of steel for automobiles. The Agricultural industry utilizes the majority of the world's water.
- Water can dissolve almost anything, this allows it to be an ideal carrier of many chemicals and minerals we need for life.
- Water is used for various household needs.
Practice problems:
Q1. In ice, there are ______ H2O molecules around each molecule of water.
A. one
B. two
C. three
D. four
Answer: C)
Solution: Each water molecule in ice is surrounded by three other water molecule, forming a hexagonal honeycomb pattern.
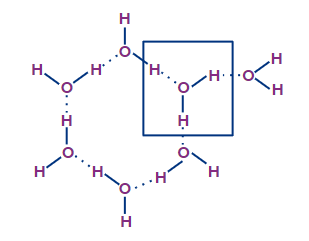
![]()
Q2. BaCl2.2H2O comes as an example of ____________.
A. Interstitial hydrates
B. Hydrogen bonded hydrates
C. Coordinated hydrates
D. catalyst
Answer: A)
Solution: BaCl2.2H2O comes as an example of Interstitial hydrates. water molecules occupy the interstitial sites in the crystal lattice. Such types of hydrates are called interstitial hydrates.
Q3. Water reacts with sulfur dioxide to give the acid as__________.
A. Hydrogen sulphide
B. Sulfurous acid
C. Sulphuric acid
D. Peroxodisulphuric acid
Answer: B)
Solution: Water reacts with sulfur dioxide to give the acid as Sulfurous acid. Below is the mentioned reaction to this.
Q4. When P4O10 reacts with water, then ____________ is produced.
A. pyrophosphoric acid
B. Phosphorous acid
C. Metaphosphoric acid
D. Orthophosphoric acid
Answer: D)
Solution: When P4O10 reacts with water, then, Orthophosphoric acid is produced. Below is the mentioned reaction to this.
Frequently asked questions- FAQs
Q1. Why does water adhere to objects?
Solution: The capacity of dissimilar molecules to bind to one another is called adhesion. With polar and charged molecules, water will form intermolecular interactions.Hydrogen bonds are held together by cohesion to provide surface tension on water. Water is drawn toward other molecules by adhesive forces because it is attracted to them.
Q2. What makes water a universal solvent, and why?
Solution: Because it can dissolve more things or substances than any other liquid, water is known as the universal solvent.
Q3. Why does wood float on water?
Solution: The sample of wood would weigh less than the sample of water if their respective volumes, or weights, were equal. Consequently, wood has a lower density than water. No matter how big or little the piece of wood is, it floats in water because it is less dense than water.
Q4. Why does water not dissolve some substances while it dissolves others?
Solution: Because they are non-polar and do not interact well with water molecules, many compounds do not dissolve in water. Oil and water are typical illustrations. Oil comprises non-polar molecules, which do not dissolve in water.



