-
Call Now
1800-102-2727
Application of Electrochemical series: Redox reactions, Electrode potential, Electrochemical series, Application of electrochemical series, Practice Problems and FAQs:
Do you like playing any sport?
Which is your favorite sport?
Well, I like playing and watching cricket. You must have seen that there’s one person in the team management who is always in front of a laptop during the match. Cricket is played with a bat and ball then what is the need of a computer guy with a computer?
Do you have any idea what is his work?
Well, his job is to provide information such as what is the strength and weakness of team members as well of the opponents on the basis of data. This information becomes very useful for the captains to take decisions during the match.
Wow, this seems fascinating isn’t it?

Similarly in chemistry, we do have a series of data that can tell us the relative activity of metals and their ions. This series can help us forecast how a reaction with a specific metal will develop in aqueous solution as well as which metal will replace another in its salt solution.
The series is referred to as electrochemical. Let's examine its fundamentals to see how valuable this series is for electrochemistry.
Table of content
- Definition of redox reaction
- Definition of Electrochemical series
- Electrode potential
- Standard electrode potentials
- Applications of Electrochemical series.
- Practice Problem
- Frequently Asked Questions (FAQs)
Definition of redox reaction
- Chemistry deals with a variety of matter and the transformation of one matter to another through various types of reactions. One important category of such reactions is the redox reactions. Any chemical reaction in which a molecule, atom, or ion's oxidation number changes as a result of gaining or losing an electron is an oxidation-reduction reaction.
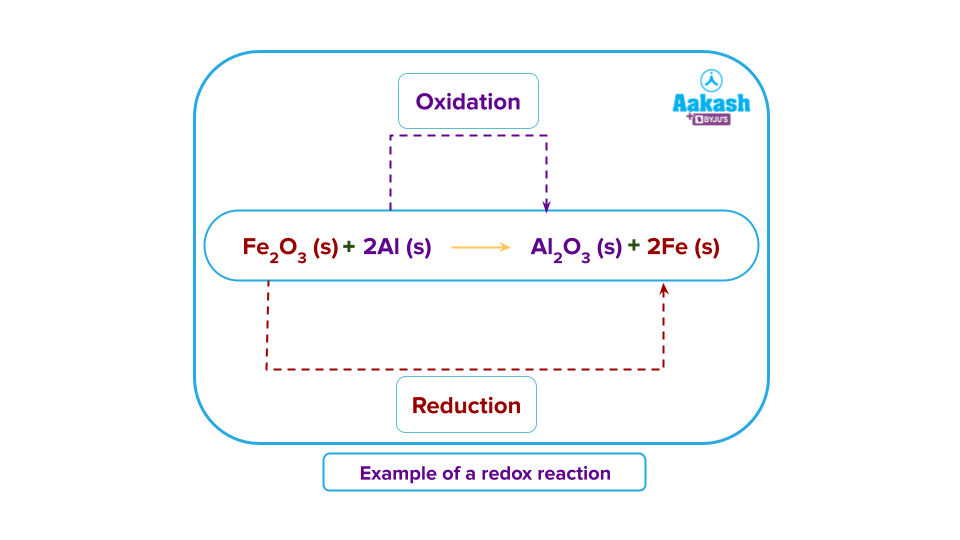
- Development of photographic plate: In photography, silver bromide is a substance found in an emulsion that aids in the development of an image. Because the grains of silver bromide are light-sensitive and suspended in gelatin, they can be used to make photographic emulsions. Silver bromide breaks down when exposed to light, which helps to preserve photographs.
Oxidation
Oxidation is the process in which a reactant either takes on oxygen or loses electrons during a reaction.
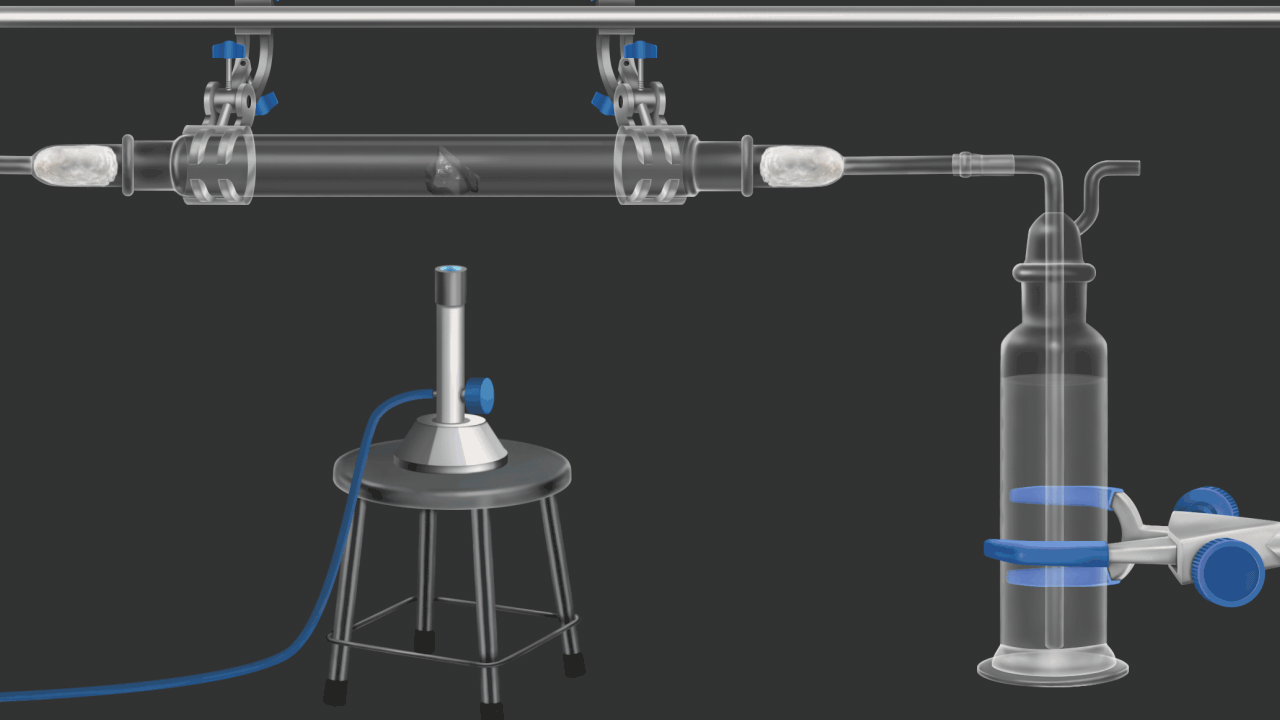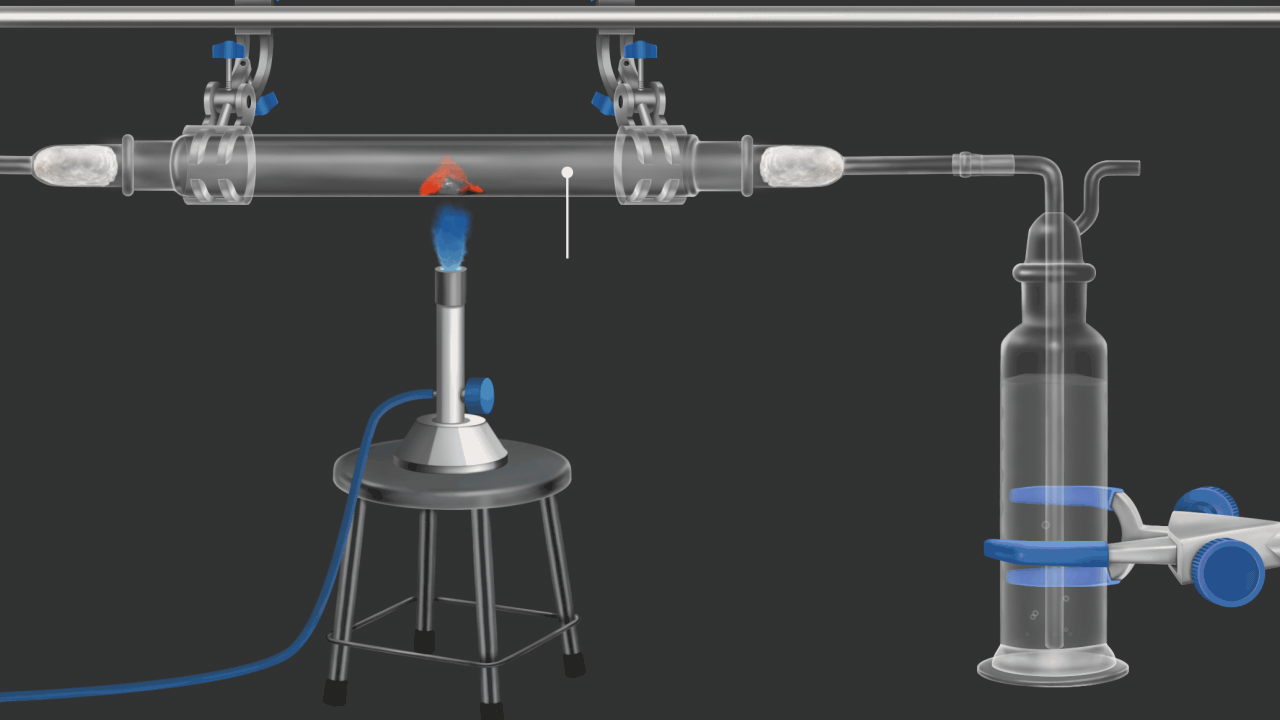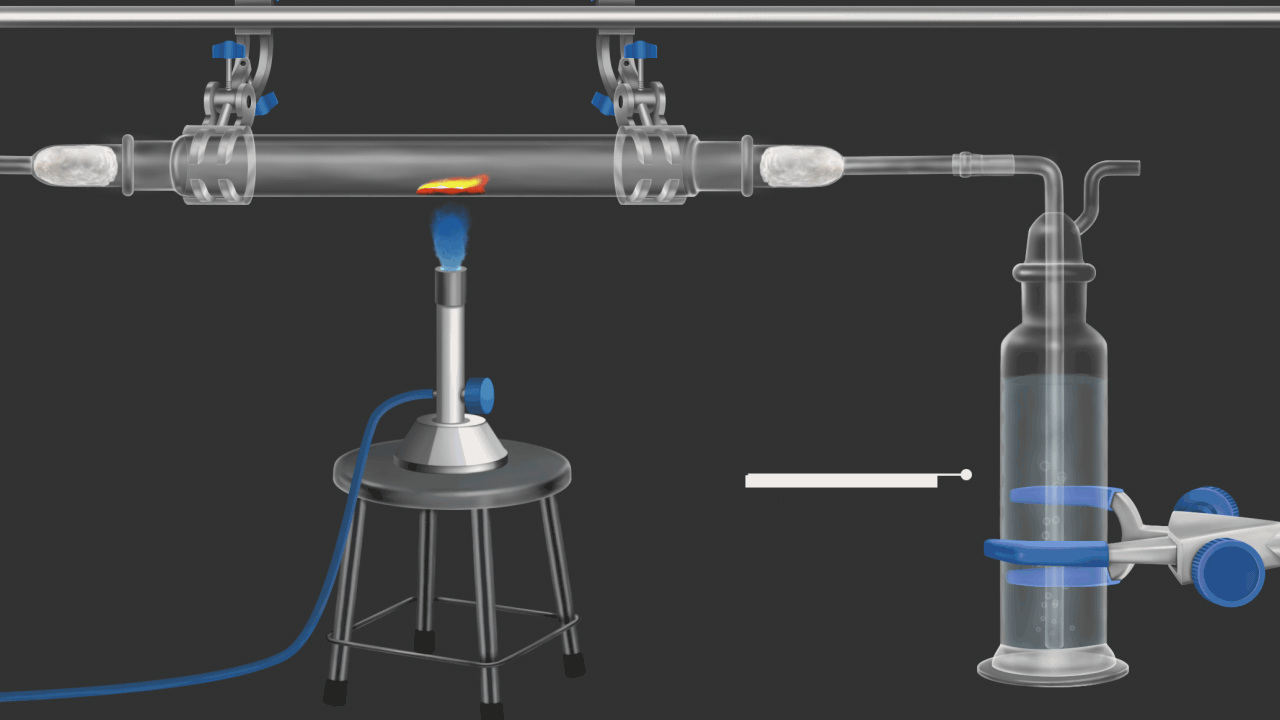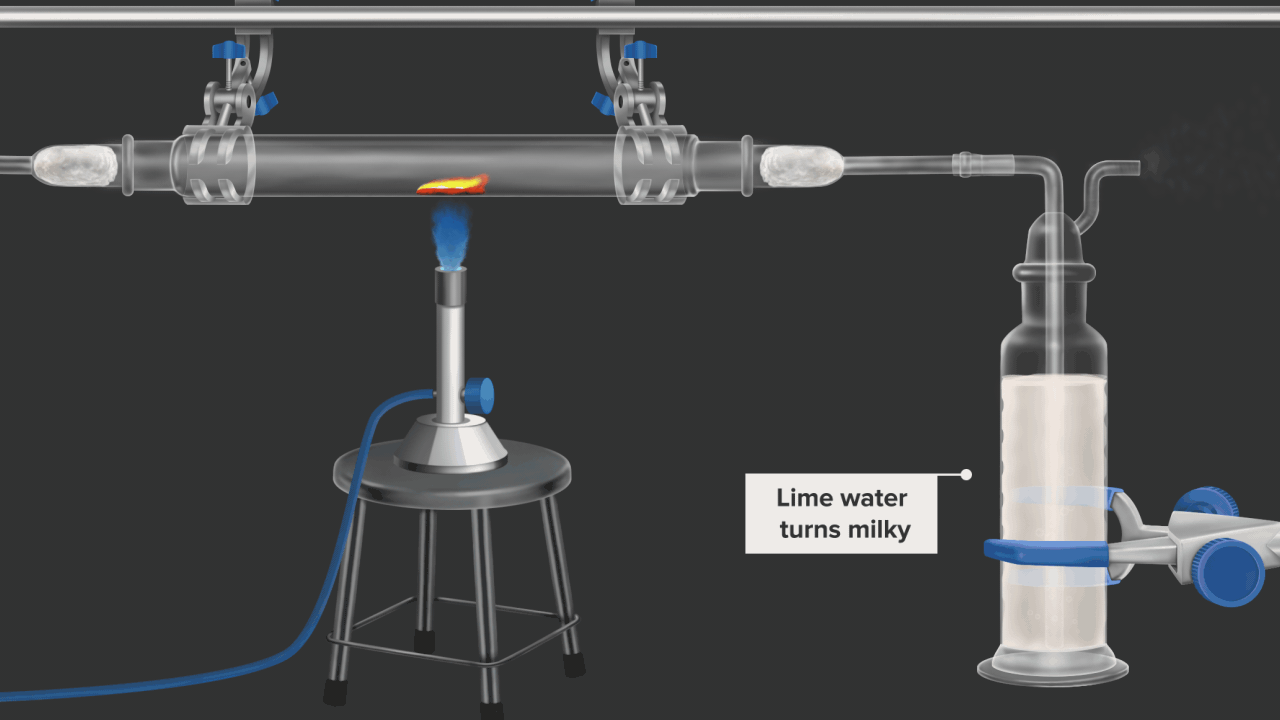
![]()
Here burning coke produces CO2gas. CO2 gas turns lime water milky. in this example oxidation of coke(carbon) is shown .
Reduction
Reduction is the term used to describe a reaction in which a reactant either lose oxygen or gains electrons.
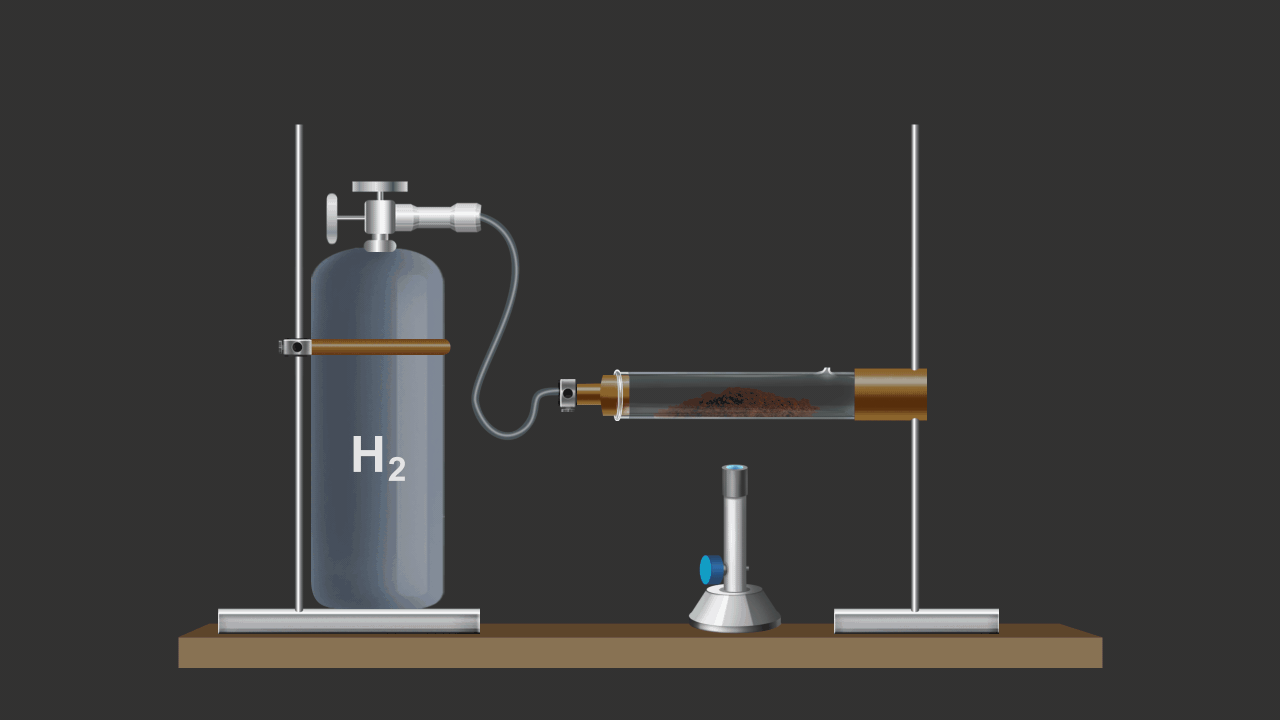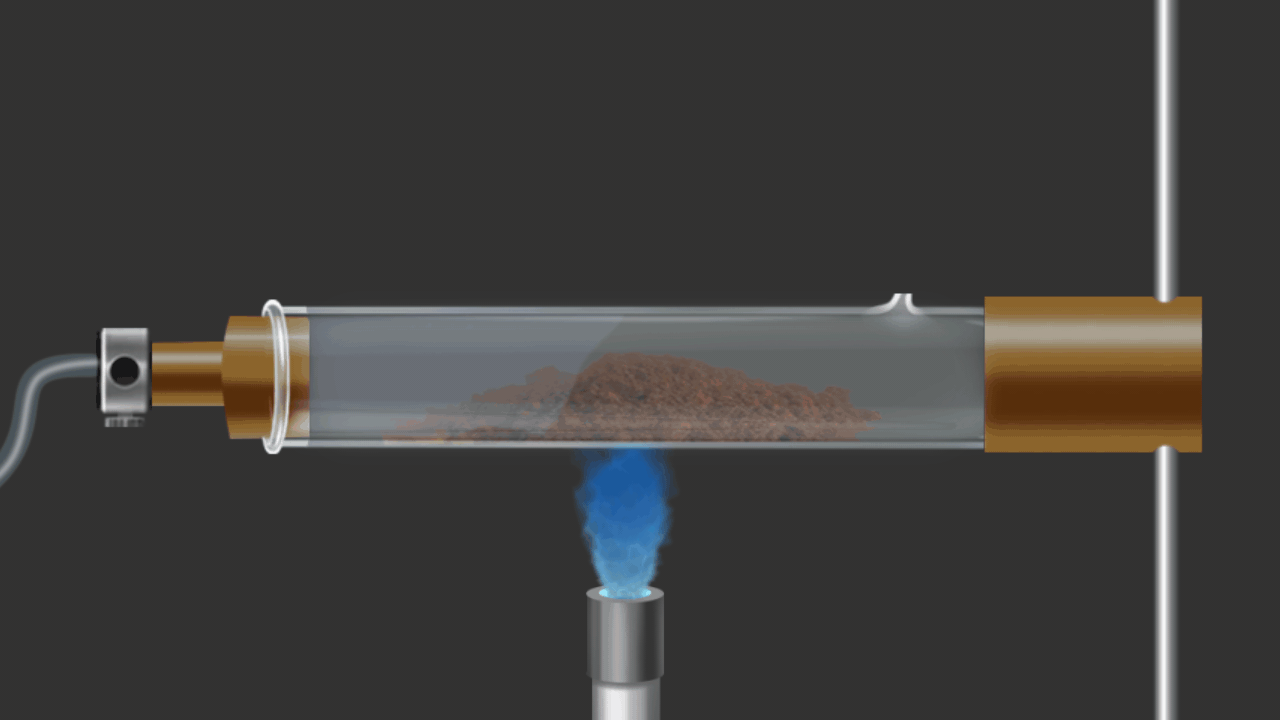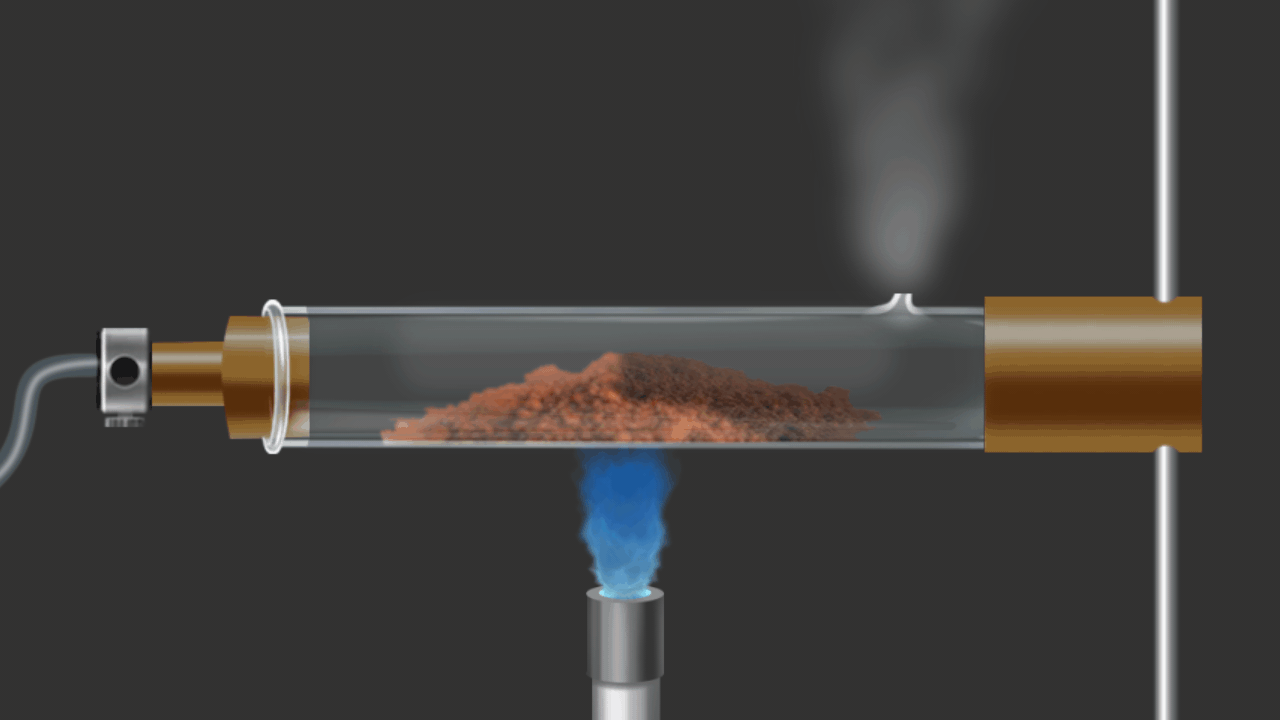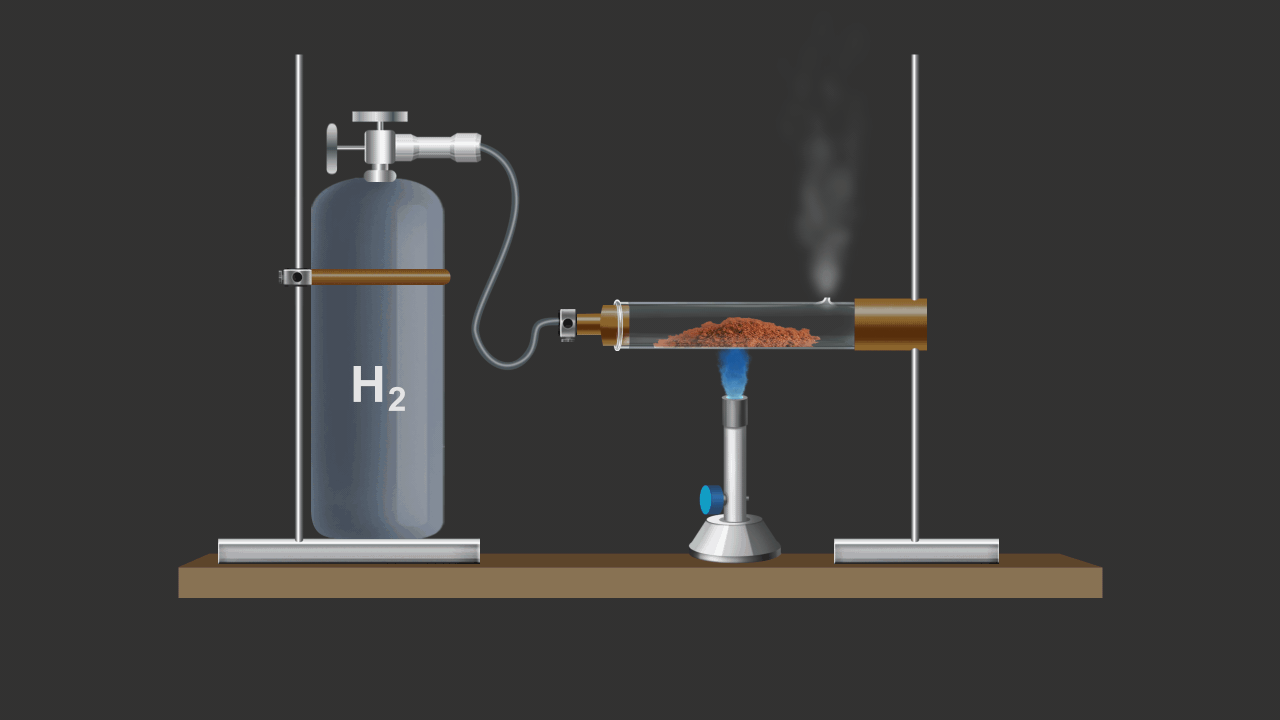
![]()
Here CuO is being reduced by addition of H2 gas on the heated oxide.
CuO (s) + H2 (g) Cu (s) + H2O (g)
Oxidizing agent
A substance or element that takes on electrons from a different species in a redox (oxidation-reduction) reaction is known as an oxidizing agent.
Reducing agent
A reducing agent is a substance that, in a redox reaction, loses electrons to other substances and undergoes oxidation to a higher valency state.
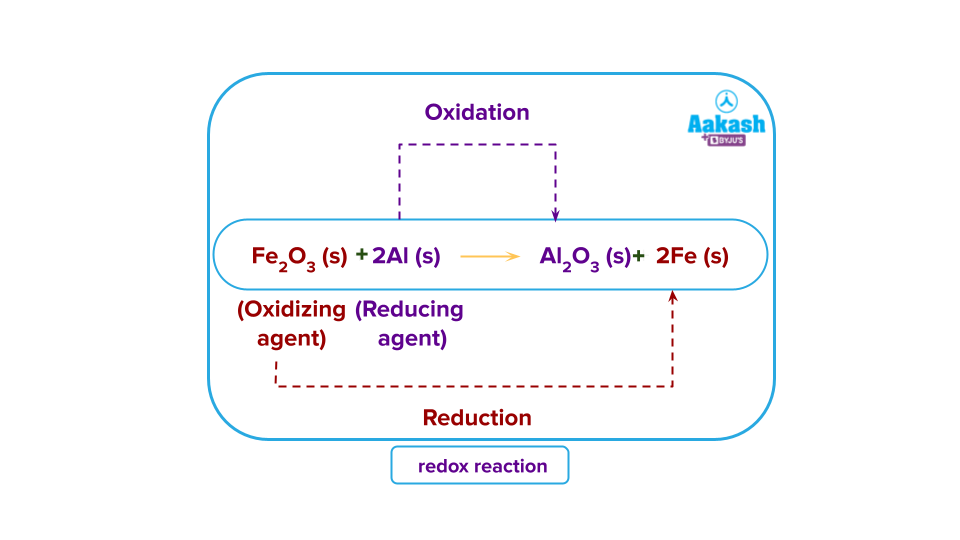
Definition of Electrochemical series
The term "electrochemical series" refers to a list that describes the arrangement of elements in ascending order of their electrode potential values. In this list, substances are arranged from higher reduction potential to lower reduction potential.
Electrode potential
According to one definition, electrode potential is the electromotive force (EMF) generated in a galvanic cell formed with the electrode in comparison to a typical reference electrode. The electrode potential is measured in volts and is indicated by the letter E. It is the measure of the ease of losing or gaining electron by a metal or ion.
Oxidation Potential
- The oxidation potential of an electrode refers to its tendency to lose electrons or become oxidized.
- The electrode potential for oxidation half-reaction
- Greater the oxidation potential, greater will be the tendency to get oxidized and lesser the tendency to get reduced. The magnitude of the oxidation potential will be the same as the magnitude of the reduction potential with the sign reversed.
Reduction potential
- The reduction potential of an electrode is the tendency of an electrode to gain or lose electrons.
- The electrode potential for reduction half-reaction
- Greater the value of reduction potential, greater will be the tendency to get reduced and lesser the tendency to get oxidized.
Standard electrode potentials
- The equilibrium potential can be measured using the standard electrode potential. The difference in potential between the electrode and the electrolyte is known as the potential of the electrode. The electrode potential is referred to as the standard electrode potential when the concentrations of all the species in a semi-cell are equal.
An electrochemical cell operates at the standard electrode potential when the following conditions are present:
temperature = 298K pressure = 1atm concentration = 1M
The symbol ''E0cell'' denotes a cell's standard electrode potential.
- The electrode potentials are frequently expressed as reduction potentials. Any electrode's reduction potential can be determined from its oxidation potential by simply changing the sign because the reduction half reaction is simply the oxidation half reaction in reverse.
- The electrode where reduction occurs in relation to a standard hydrogen electrode has a reduction potential that is given a positive sign, and the electrode where oxidation occurs in relation to a standard hydrogen electrode has a positive oxidation potential or, when expressed as reduction potential, a negative sign.

Applications of Electrochemical series.
1. Comparing various metal’s and their ion’s oxidising and reducing properties.
- As is usual, a positive sign is used to denote the reduction potential. A higher reduction potential indicates that the substance (element or ion) will be reduced more readily, or that it is a stronger oxidizing agent.
- F2 is reduced most readily whereas lithium ions are reduced most difficultly because F2has the highest reduction potential and Li+ ion has the lowest. To put it another way, F2 is the most potent , whereas Li+ ions are the least potent oxidizing agent.
- In contrast, if the reaction is written in reverse order, it can be postulated that lithium will be the most easily oxidized and is therefore the best reducing agent, whereas fluoride ions will be the most difficult to oxidize and are therefore the weakest reducing agents.

2. The relative activities of metals can be compared.
A metal's oxidation potential determines how easily it can lose electrons and how reactive it is. Because of this, metals with higher oxidation potentials have the ability to remove metals with lower oxidation potentials from salt solutions. For instance, the order of the oxidation potentials of Mg, Zn, Fe, Cu, and Ag:
Mg>Zn> Fe > Cu > Ag
As a result, each metal can displace metals below from salt solutions. As a result, their reactivity is in the preceding order.
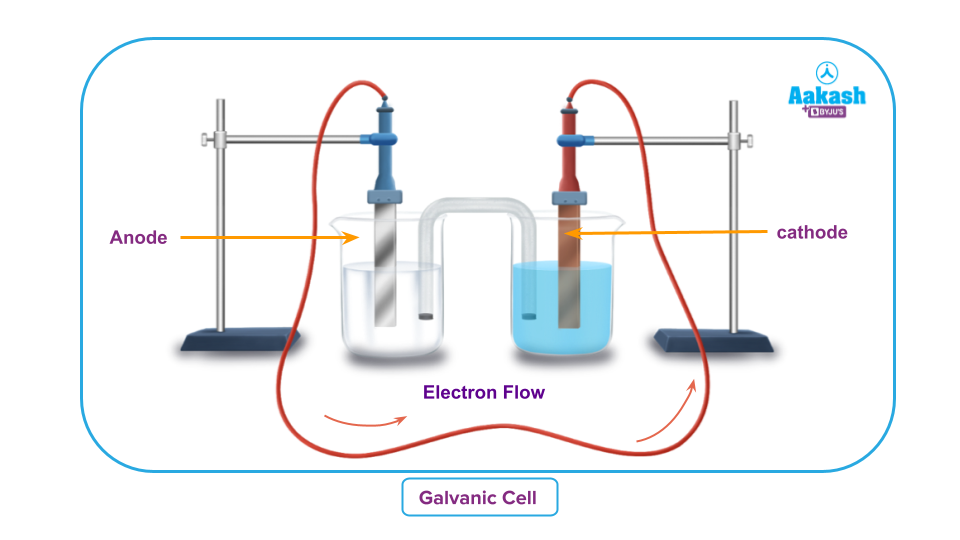
3. Determining the standard EMF of any galvanic cell.
- A galvanic cell or voltaic cell is an electrochemical device that transforms the chemical energy of spontaneous redox reactions into electrical energy. The reaction taking place in a galvanic cell can be represented by two half cell reactions that is
- Oxidation half reaction
- Reduction half reaction
std EMF of the cell=Std. oxdnpotential of the oxdnhalf reaction+std. rednpotential of the redn half reaction
As
Oxidation potential = - Reduction potential,
the above expression may also be written in the form :
std EMF of the cell=Std. rednpotential of the oxdnhalf reaction+std. oxdn potential of the redn half reaction
Additionally, just like in a representation of a cell, the electrode where oxidation occurs is written on the left, and the electrode where reduction occurs is written on the right.
E0cell=E0cathode- E0anode
4. To determine if a metal will produce hydrogen gas when it reacts with an acid.
Previously we all have learnt that metals when reacted with an acid produces H₂ gas. Not all metals, but a lot of them, react with acids. As the metal and acid combine to form a salt, hydrogen gas is produced.
The following reaction must occur in order for a metal A (assuming it is monovalent) to react with an acid to produce H2gas:
A+H+ A++ 12H2
AA++e-
H++e-12H2
As a result, the metal ought to have a tendency to lose electrons, or to experience oxidation, in the presence of hydrogen. As a result, in the electrochemical series, all metals above hydrogen react with the acid to produce hydrogen gas. Furthermore, it is obvious that higher reactivity results from lower reduction potential (i.e., more negative reduction potential).
5. Predicting the redox reaction's spontaneity.
The EMF of the cell based on the given redox reaction is calculated to determine whether a given redox reaction is possible or not. To happen spontaneously, a redox reaction needs the cell's EMF to be positive. If the EMF is found to be negative, the given direct reaction cannot occur; however, the opposite reaction may occur.
6. To determine the minimal voltage necessary to electrolyze a salt solution.
When we carry out an electrolysis of a salt solution, we apply some potential difference across the cell. One thing we have to remember is that not every salt solution will carry out electrolysis at the same potential difference. Hence, it is important to know what will be the minimum amount of voltage that can be applied to proceed electrolysis. This data can be obtained with the help of electrochemical series. We can simply write down the reaction at anode and cathode. By obtaining the overall net reaction of cell, minimum voltage can be calculated.
Practice Problem
Q.1 Determine the standard EMF of a cell using electrochemical series which has cell reaction as
Zn+2Ag+ Zn2++2Ag
E0Zn,Zn2+=0.76 volt
E0Ag,Ag+= -0.80 volt
- 1.56 volts
- 15.6 volts
- 0.156 volts
- 0.0156 volts
Answer: (A)
Solution: Let's first divide the aforementioned reaction into two half-cell reactions.
Zn Zn2++2e- (Oxidation)
2Ag++2e- 2Ag (reduction)
If you look at the information above, it represents the standard oxidation potential of both silver and zinc.
We need the reduction potential of the silver electrode and the oxidation potential of the zinc electrode to calculate the standard EMF of the cell.
Reduction potential of Ag electrode = -Oxidation potential of Ag electrode
= -(-0.80 volt)= +0.80 volt
Standard EMF of the cell = Standard oxidation potential of Zn electrode + Standard reduction potential of Ag electrode = 0.76+0.80 volt=1.56 volts
Q.2 What will happen if a copper metal block is fallen into a beaker containing a 1M ZnSO4 solution?
- Copper metal will be dissolved in the solution and N2 gas will evolve.
- Copper metal will be dissolved in the solution and H2 gas will evolve.
- No reaction will take place
- Copper metal will be dissolved in the solution and O2 gas will evolve
Answer: (C)
Solution: Metals with lower reduction potentials can be displaced from their salt solution by metals with higher reduction potentials.
The following is the order of the metal's reduction potential.
Mg<Zn< Fe <Cu <Ag
Hence, Cu can’t displace Zn from its salt solution.
Q.3 Which of the following statements will come true if zinc and silver react with 1M sulphuric acid.
E0Zn,Zn2+=0.76 volt
E0Ag,Ag+= -0.80 volt
- reaction of Zn and Ag with sulphuric acid takes place to give hydrogen gas.
- reaction of Zn with sulphuric acid takes place to give hydrogen gas.
- reaction of Ag with sulphuric acid doesn’t take place to give hydrogen gas.
- Both B & C
Answer: (D)
Solution:
(i) reaction of Zn with sulphuric acid
Zn+H2SO4ZnSO4+H2
Cell representation will be : Zn|Zn2+||H+|H2
Standard EMF of the cell = Standard reduction potential of R.H.S electrode - Standard reduction potential of L.H.S. electrode
=0-(-0.76)=+0.76 volt
EMF is positive. Hence, reaction of Zn with sulphuric acid takes place.
(ii) reaction of Ag with sulphuric acid
2Ag+H2SO4ZnSO4+H2
Cell representation will be : Ag|Ag+||H+|H2
Standard EMF of the cell = Standard reduction potential of R.H.S electrode - Standard reduction potential of L.H.S. electrode
= 0-0.80= -0.80 volt
EMF is negative. Hence, reaction of Ag with sulphuric acid doesn’t take place.
Q.4 What is the minimum voltage required to bring about the electrolysis of 1M copper sulphate solution at 298 K.
E0Cu,Cu2+=0.34 volt
E0H2O,H+= -1.23 volt
- 8.9 V
- 9.8 V
- 0.89 V
- 0.98 V
Answer: (C)
Solution:
Anode: Cu2++2e-Cu, E0=0.34 volt
Cathode: H2O2H+ +12O2+2e-, E0= -1.23 volts
Net Reaction: Cu2++H2OCu+12O2+2H+, E0cell= -0.89 volts
Minimum voltage required for the electrolysis is 0.89 V
Frequently Asked Questions (FAQs)
Q.1 Are EMF and potential difference same?
Answer: No, these two terms are different from each other.EMF is the potential difference between the two electrodes of the cell when no current is flowing in the circuit. Potential difference is the difference in the electrode potentials of the two electrodes under any condition.
Q.2 Why is hydrogen's electrode potential assumed to be zero?
Answer: Without first forming a complete cell, it is impossible to determine an electrode's absolute electrode potential. In light of this, hydrogen is selected as the common reference electrode with a zero electrode potential.
Q.3 Why is it impossible to determine an electrode potential's absolute value?
Answer: Electrode potentials cannot be measured in absolute terms because oxidation and reduction cannot occur in isolation. Consequently, an electrode's potential is measured using a reference electrode.
Q.4 Can electrochemical series be used to predict reaction rate?
Answer: The electrochemical series has another drawback in that it cannot provide information on the rate of half-reaction reactions. Even though one reaction is predicted to happen before another, that doesn't mean it will in fact happen.



