-
Call Now
1800-102-2727
Uses of Ammonia - Ammonia, Important Properties, Important Uses and Applications, Practice Problems and FAQ
You've probably seen beautiful women advertising for various hair-dye brands during commercial breaks, claiming that their products are "Ammonia-free," haven't you?

In fact, this has become a cliche statement for a brand supporter or influencer attempting to promote a hair-colouring product brand. But, as it is a very important component in the production of hair dye, it is difficult to create one that is completely free of it! Ammonia is a multifaceted chemical that is extremely popular in both the industrial and agricultural sectors. It is a significant chemical that has served as a link between these two industries, which some regard are opposites.
They do reciprocate, believe me! But that is not the topic of discussion today. Leaving that aside, let us learn more about the major applications and significance of ammonia, specifically what it has to offer.
TABLE OF CONTENTS
- Ammonia - Introduction
- Ammonia - Structure
- Ammonia - Properties
- Ammonia - Uses
- Practice Problems
- Frequently Asked Questions - FAQ
Ammonia - Introduction
Ammonia is a colourless alkaline gas that has a strong pungent odour. It can be found in the environment, combined in the air, soil, or water. It can be found in plants, animals, and even humans.
Furthermore, when hydrogen and nitrogen react, ammonia, also known as azane, is created. The chemical formula for it is NH3. It has high water solubility. Apart from that, after sulphuric acid, ammonia is the world's second most manufactured material in terms of volume. Other than that, in terms of volume, the second most manufactured chemical in the world is ammonia.
Ammonia - Structure
Ammonia is obtained from nitrogen and hydrogen. The atomic number of nitrogen is 7 and its electronic configuration is 1s2 2s2 2p3. Ammonia undergoes sp3 hybridisation and also there is the presence of a lone pair, so ammonia is trigonal pyramidal in shape.
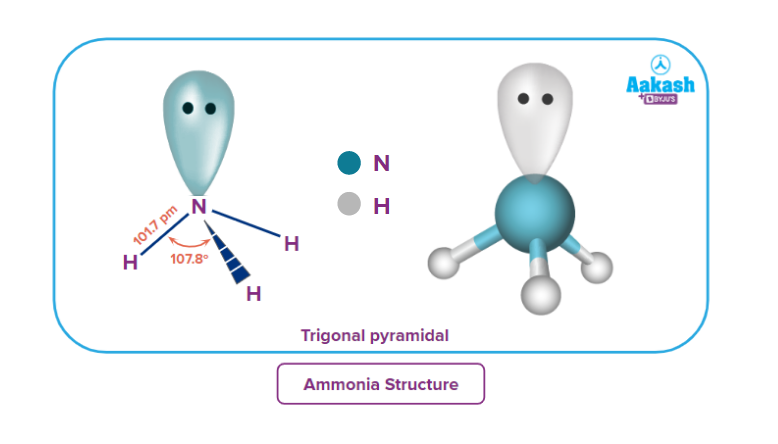
Ammonia - Properties
- Ammonia is a colourless, very unpleasant gas with a terrible, stifling stench at room temperature. At room temperature, ammonia has a stifling stench, is very unpleasant and colourless.
- It is hygroscopic and is known as anhydrous ammonia in its pure state (readily absorbs moisture). In the pure state, it readily absorbs moisture as it is hygroscopic; it is known as anhydrous ammonia.
- Ammonia is corrosive and has alkaline characteristics. Ammonia is alkaline in nature and gas corrosive properties.
- Ammonium hydroxide, a caustic solution and weak base is formed when ammonia gas dissolves easily in water. When ammonia is dissolved in water, a caustic solution called ammonium hydroxide is obtained.
- Under pressure, ammonia gas compresses easily and becomes a transparent liquid. Ammonia gas can be compressed easily; it becomes a transparent liquid under high pressure.
- Ammonia is frequently transported in steel containers as a compressed liquid. Generally, ammonia is transported as a compressed liquid in steel containers.
- Although ammonia is not very flammable, it can explode when subjected to tremendous heat. Though ammonia is not flammable, when exposed to significant amounts of heat, it can explode.
- In the solid and liquid states, it is associated via hydrogen bonds. Ammonia molecule is polar due to strong intermolecular hydrogen bonding.
- It has a dielectric constant of 22 at −34 °C (−29 °F) which is lower than that of water. Hence, it serves better as a solvent for all the organic materials.
- Ammonia acts as a base as it has a lone pair of electrons which can be donated. Hence, it is a Lewis base.
Ammonia - Uses
Ammonia in Agricultural Sector
- Ammonia is a fundamental component of ammonium nitrate fertiliser, which that releases nitrogen, an important nutrient for growing plants such as farm crops and lawns a nutrient that is significant for growing plants such as lawns and farm crops.
- Around 90% of the ammonia produced globally is utilised in making nitrogenous fertiliser,. which helps to ensure food production for billions of people. For billions of people around the world, this ensures food production.
- The production of food crops Food crop production depletes soil nutrient levels in a natural way. Farmers rely depend on fertiliser to make their soils productive productive soils in order to produce healthy crops.
- Fertilisers can also assist food crops that have higher amounts of critical elements including zinc, selenium, and boron boron, zinc and selenium.
- Ammonia also functions as an antifungal agent on certain fruits and acts as preservatives.
Ammonia in Household and Cleaning products
- Many household cleaning solutions contain ammonium hydroxide, often known as household ammonia, which is used to clean a variety of surfaces such as tubs, sinks, toilets, countertops, and tiles that can be utilised to clean various surfaces like tiles, sinks, tubs, countertops and toilets.
- Using ammonia, Hhousehold dirt or stains from animal fats or vegetable oils, such as cooking grease and wine stains can also be broken down using ammonia vegetable oils or animal fats like wine stains and cooking grease can be also broke down..
- In solutions used to clean glass, ammonia is used Ammonia is widely used in glass cleaning solutions to assist reduce streaks because it dissipates quickly.
- Ammonia can also be utilised as an antibacterial or antiseptic agent, as well as a fuel.
Ammonia in Manufacturing Other Chemical Compounds
- Ammonia is used to make a variety of products, including nitric acid, hydrogen cyanide, ammonium carbonate, phenol, urea, amino acids, ammonium nitrate, ammonium phosphate, ammonium sulphate, urea, and many others.
- Hydrazine made from the reduction of ammonia with hydrogen has great potential to serve as rocket fuel.
- Ammonia is useful in several industrial processes like in manufacturing nitric acid by Ostwald’s process and in manufacturing sodium carbonate by Solvay’s process.
Ammonia in Industrial use
- From its surrouundings, aAmmonia may absorb a lot of heat from its surroundings, when employed as refrigerant gas and in air-conditioning equipment.
- Liquid ammonia acts as a refrigerant.
- Ammonia is utilised in water prurification Ammonia is used to purify water and as a component in the production of a wide range of products, including plastics, explosives, textiles, insecticides, and colours products like insecticides, colours, explosives, plastics and textiles.
- Ammonia is also utilised as a stabiliser, neutralizer, and source of nitrogen in the waste and wastewater treatment, cold storage, rubber, pulp and paper, and food and beverage sectors in cold storage, pulp, rubber, paper, beverage and wastewater treatment sectors. It is also used in the pharmaceutical industry.
Ammonia in Petroleum and Mining
- Ammonia is used in the petroleum industry to balance the acid elements of crude oil. It also aids in the prevention of corrosion in equipment. Ammonia is also utilised in the mining industry to recover a variety of metals.
Ammonia in Metal Treatment
- Carbo nitriding, nitriding, furnace brazing, brilliant annealing, sintering, atomic hydrogen welding, and other such processes use dissociated ammonia. Also, in the extraction of various metals, it finds utility.
Practice Problems
Q. 1. While using cleaning products containing ammonia, disclaimers like “Do not mix bleach with ammonia” are written. Why so?
Answer: Mixing ammonia with chlorine bleach chlorine bleach with ammonia is particularly dangerous because deadly fumes known as chloramines are produced. Coughing, shortness of breath, chest pain, nausea, irritation of the throat, nose, and eyes, or pneumonia and fluid in the lungs Shortness of breath, coughing, nausea, chest pain, irritation of the nose, throat, and eyes, and fluid in the lungs or pneumonia can all be symptoms of chloramine gas exposure.
Chloramines are basically NH2Cl, NHCl2 and NCl3. Uncontrolled evolution of these gases occurs in a chain-like reactions that can be harmful.
Q. 2. In household cleaning applications, ammonia is used as
- Antifungal Agent
- Antimicrobial Agent
- Antiseptic Agent
- All of the above
Answer: Ammonia has the inherent characteristics of controlling all kinds of microbial growth and cleaning surfaces. Hence, it acts as all of the above.
So, option D) is the correct answer.
Q. 3. Ammonia is
- A Lewis acid
- A Lewis base
- Amphoteric in nature
- None of the above
Answer: Ammonia acts as a Lewis base as it has a lone pair of electrons which can be donated. Hence, it is a Lewis base.
So, option B) is the correct answer.
Q. 4. Name the product obtained from ammonia that is used in making rocket fuels.
- Chloramines
- Azane
- Hydrazine
- Azobenzene
Answer: Hydrazine (N2H4) obtained by the reaction of ammonia with hydrogen is used as rocket fuel.
So, option C) is the correct answer.
Frequently Asked Questions - FAQ
1. What are the natural sources of ammonia?
Answer: Decomposition or breakdown of organic waste matter, gas exchange with the atmosphere, forest fires, animal and human waste, and nitrogen fixation activities are all natural sources of ammonia. Natural sources of ammonia include the breakdown of organic waste materials, atmospheric gas exchange, fires in the forest, human and animal waste, and nitrogen fixation processes.
2. How does ammonia affect aquatic life?
Answer: Ammonia can enter the aquatic environment through both direct and indirect sources, including municipal effluent discharges and animal excretion of nitrogenous wastes, as well as nitrogen fixation, air deposition, and runoff from agricultural fields.
Due to the presence of excessive ammonia in water, aquatic creatures struggle to evacuate the toxicant, which can lead to toxic buildup in internal tissues and blood, as well as mortality. Ammonia toxicity in aquatic animals can be affected by environmental conditions such as pH and temperature. Both direct and indirect sources, such as municipal effluent discharges, animal excretion of nitrogenous wastes, nitrogen fixation, air deposition, and runoff from agricultural fields, can introduce ammonia into the aquatic environment.
Water that contains too much ammonia makes it difficult for aquatic life to expel the poison, which can result in harmful accumulation in internal tissues and blood as well as mortality. Environmental factors like pH and temperature can have an impact on an aquatic animal's susceptibility to ammonia poisoning.
3. How is ammonia formed in the human body?
Answer: When the body breaks down the proteins in the food that we intake, the proteins get converted into amino acids and ammonia. This by-product of ammonia gets subsequently converted to urea and is excreted through urine. The proteins in the food we eat are broken down by the body into amino acids and ammonia by the time we eat. This ammonia byproduct is eventually transformed to urea and eliminated by the urine.
4. How can we be exposed to ammonia?
Answer: Because ammonia occurs naturally and is also found in cleaning products, exposure from these sources is possible. Due to the extensive use of ammonia on farms, in industrial and commercial settings, exposure might occur as a result of an unintentional release. Ammonia is a naturally occurring substance that can also be present in cleaning supplies, therefore exposure from these sources is conceivable. Due to the widespread usage of ammonia in industrial, commercial, and agricultural contexts, exposure could happen from an unintended discharge.
Because anhydrous ammonia gas is lighter than air and rises, it dissipates quickly and does not settle in low-lying locations. Anhydrous ammonia gas quickly dissipates and does not settle in low-lying areas since it is lighter than air and rises. The liquid anhydrous ammonia gas, on the other hand, creates vapours that are heavier than air in the presence of moisture (such as high relative humidity). Contrarily, in the presence of moisture, liquid anhydrous ammonia gas produces vapours that are heavier than air (such as high relative humidity). People may be exposed to these fumes if they spread along the ground or into low-lying locations with limited airflow.
5. What are the harmful impacts of ammonia exposure?
Answer: Ammonia is an irritant and corrosive substance. When exposed to high levels of ammonia in the air, the nose, throat, and respiratory tract become immediately burned. Bronchiolar and alveolar oedema, as well as airway damage, might result in respiratory distress or failure.
Low amounts of ammonia in the air or solution can cause immediate skin or eye discomfort. Ammonia concentrations above a certain level can cause serious harm including burns. Swallowing ammonia solution exposes the lips, throat, and stomach to high quantities of ammonia, which causes corrosive damage. A caustic and irritating material is ammonia. The nose, throat, and respiratory system become quickly burned when exposed to high quantities of ammonia in the air. Respiratory discomfort or failure may be caused by airway injury, bronchiolar and alveolar oedema, or both.
Low concentrations of ammonia in the atmosphere or solution can immediately irritate the skin or eyes. Any major harm, including burns, can be brought on by ammonia concentrations above a particular point. The lips, throat, and stomach are exposed to large amounts of ammonia when you swallow ammonia solution, which damages them by being corrosive.



