-
Call Now
1800-102-2727
Preparations of alcohol: Classification, Preparations, Industrial preparations, Practice problems and Frequently asked questions(FAQs)
Do you know what chemical is present in biofuel?

It is ethanol that falls within the category of alcohol.
Biofuels are being looked at more and more as potential replacement fuels for the future because fossil fuels are non-renewable and contribute to global warming.
Because ethanol burns completely, it serves as a biofuel. As a result, it does not significantly increase pollutants. It produces water and carbon dioxide while it burns with a clear, smokeless flame.
Typically, ethanol is combined with gasoline to create "gasohol," which contains around 10% ethanol.
Our reliance on naturally occurring fossil fuels like gasoline is decreased when we use ethanol as a fuel. Sugar cane is widely grown in nations like Brazil that have scarce natural oil resources. To make ethanol, sugar cane must be fermented. The ethanol produced by fermentation is utilised as a fuel substitute.

Though as per chemistry Alcohol is a solution, it should not be used in the name of solution for your problems in life.
So teenagers should be aware of alcohol. It can be the reason for your health problems but it can never be the solution for your current problems.
Table of content:
- Classification of alcohols
- Preparations of alcohols
- Industrial preparations of alcohols
- Practice problems
- Frequently asked questions(FAQs)
Classification of Alcohol:
Classification of Alcohols on the basis of -OH groups:
Alcohols are classified into four categories based on the number of hydroxyl groups attached to the carbon atoms: Monohydric alcohol, dihydric alcohol, trihydric alcohol and polyhydric alcohol.
Alcohol is classified as follows:
1. Monohydric alcohol: Monohydric alcohol is alcohol that contains only one hydroxyl group.
Methyl alcohol, ethyl alcohol, and propanol are some examples.
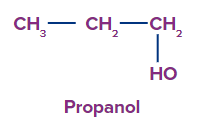
2. Dihydric alcohol: Dihydric alcohol is a type of alcohol that contains two hydroxyl groups.
For example-1,2–Ethanol, 1,3 propandiol.
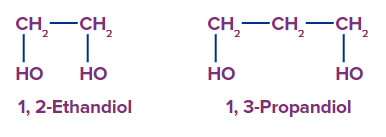
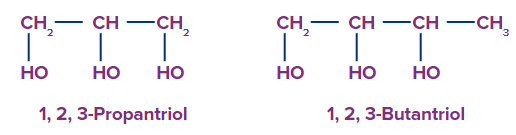
3. Trihydric alcohol: Trihydric alcohol is alcohol with three hydroxyl groups.
For example-1,2,3-Propantriol, 1,2,3-Butanetriol.
4. Polyhydric alcohol: Polyhydric alcohol is defined as alcohol that contains four or more hydroxyl groups.
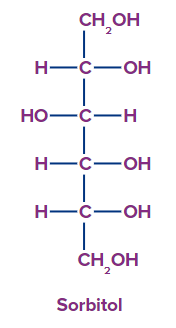
Alcohol classification based on the nature of the carbon atom bonded to the -OH group:
Alcohols are classified into three types based on the number of carbon atoms directly attached to the carbon bonded with the -OH group: primary, secondary, and tertiary.
1. Primary alcohols: primary alcohols have only one carbon atom along with the -OH group attached to it.
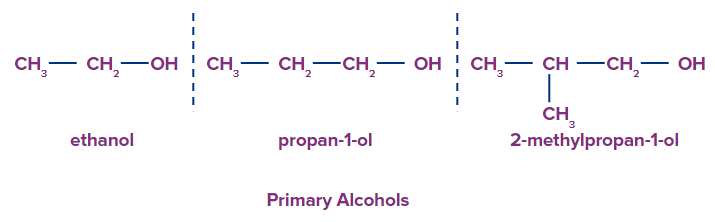
2. Secondary alcohols: Secondary alcohols have two carbon atom along with the -OH group attached to it.
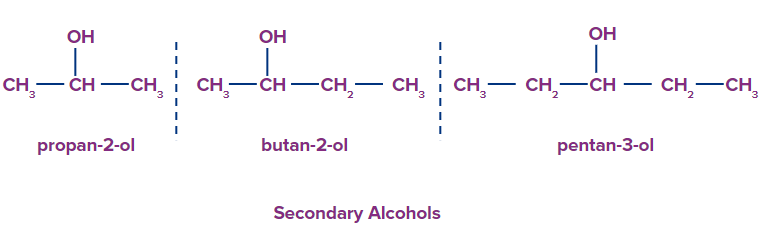
3. Tertiary alcohols: Tertiary alcohols have three carbon atom along with the -OH group attached to it.
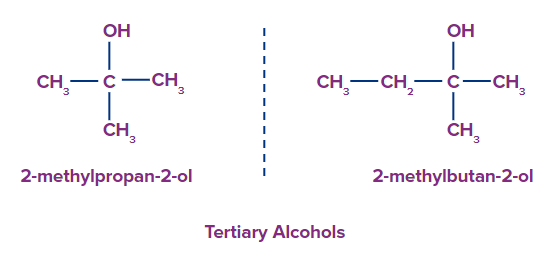
Preparations of Alcohols:
1. Acid catalysed hydration:
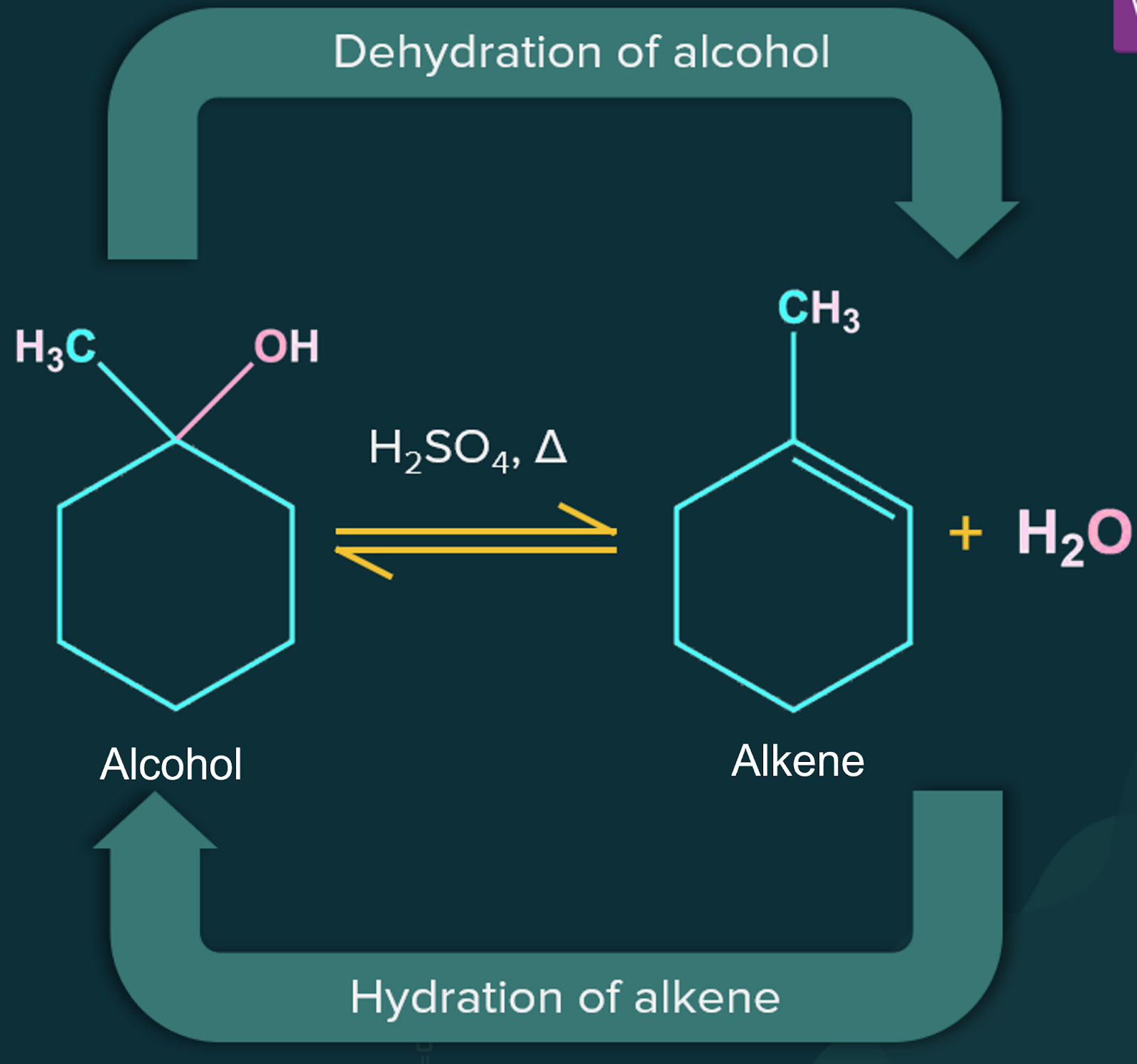
Alcohols can be produced by hydrating the alkenes. Indirect hydration of alkenes can be accomplished by adding sulphuric acid followed by hydrolysis with hot water to produce alcohol. Acid-catalysed hydration proceeds through Markovnikov’s rule for unsymmetrical alkenes.
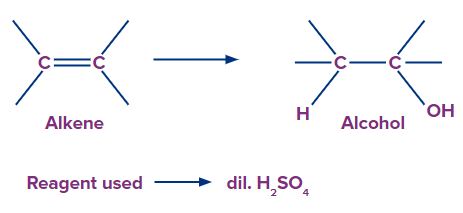
Acid catalysed hydration of alkenes is reversible and the mechanism of acid catalysed hydration of alkenes is simply the reverse of that for the dehydration of alcohol.
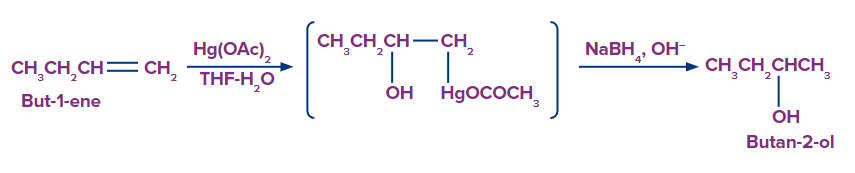
2. Hydration by Oxymercuration-Demercuration:
Alkenes can be converted to alcohol using the oxymercuration-demarcation reaction. An alkene is treated with mercuric acetate in a THF–water solution to produce a mercurated product, which can then be reduced with NaBH4 in the basic medium to produce alcohol. According to Markovnikov's rule, addition occurs in this reaction.
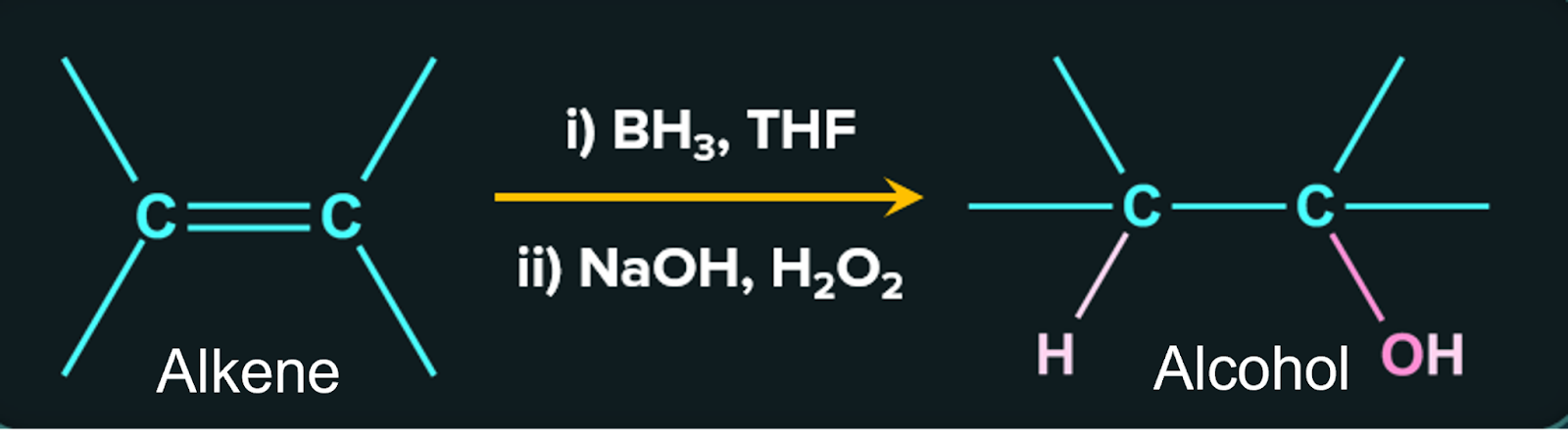
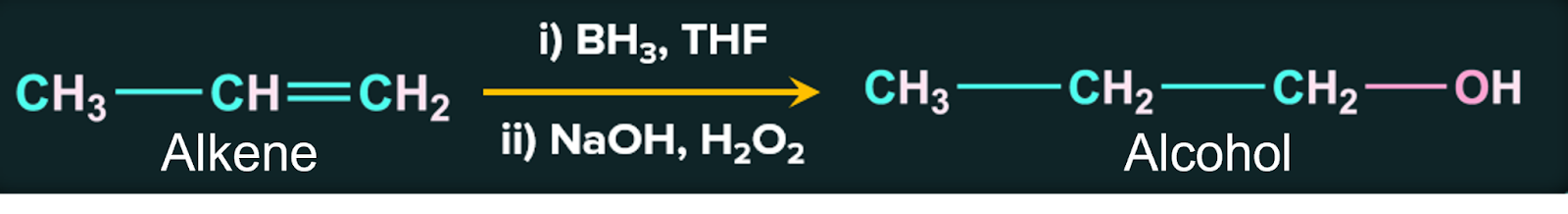
3. By the hydroboration – oxidation reaction:
The hydroboration – oxidation reaction can also add water to olefins indirectly. When an alkene is treated with diborane BH3/THF and then with H2O2 in the presence of aqueous sodium hydroxide, alcohol is produced. Diborane acts as an electrophile in this process because it is an electron deficient compound.
Hydroboration-oxidation reactions are regioselective. The net result of hydroboration-oxidation is anti-markovnikov addition of water to an alkene in case of unsymmetrical alkenes.
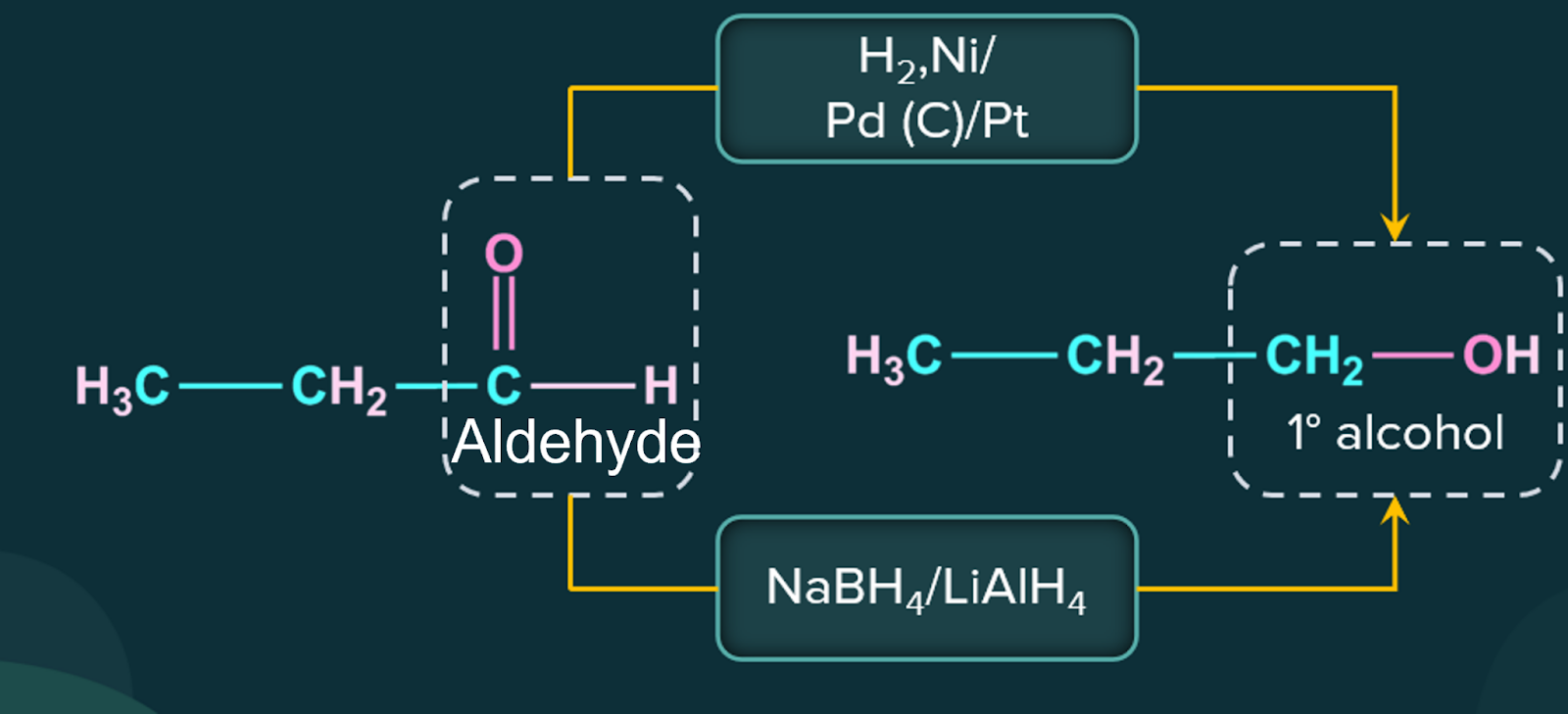
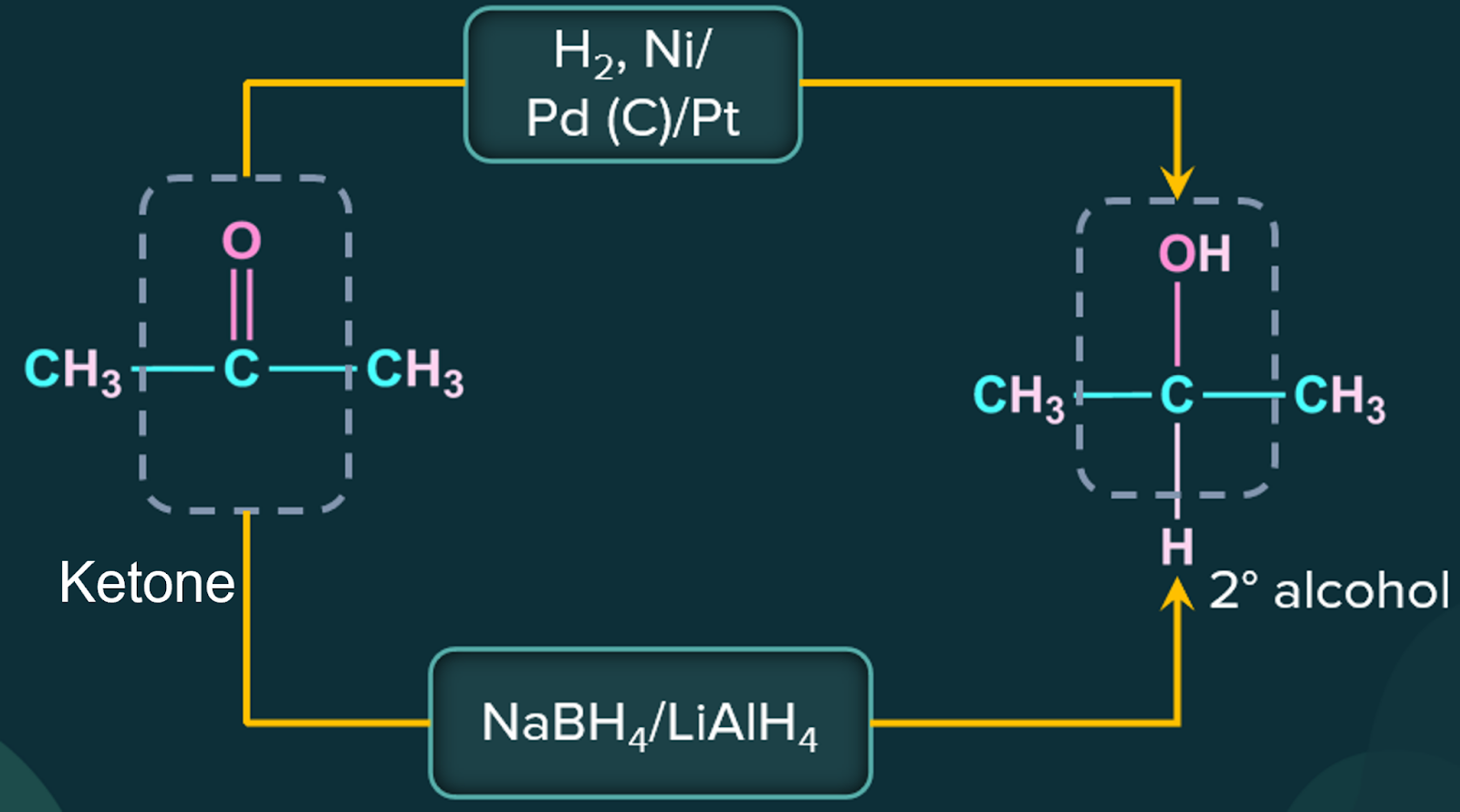
4. Preparation of Alcohol by Reduction of Aldehydes and Ketones:
Alcohols are produced by reducing aldehydes and ketones. Common reducing agents perform the following reduction processes:
(i) Hydrogen with a catalyst such as finely divided platinum, palladium, nickel, or ruthenium.
(ii) Lithium aluminium hydride or sodium borohydride.
Aldehyde produces primary alcohols during reduction, whereas ketone produces secondary alcohols.
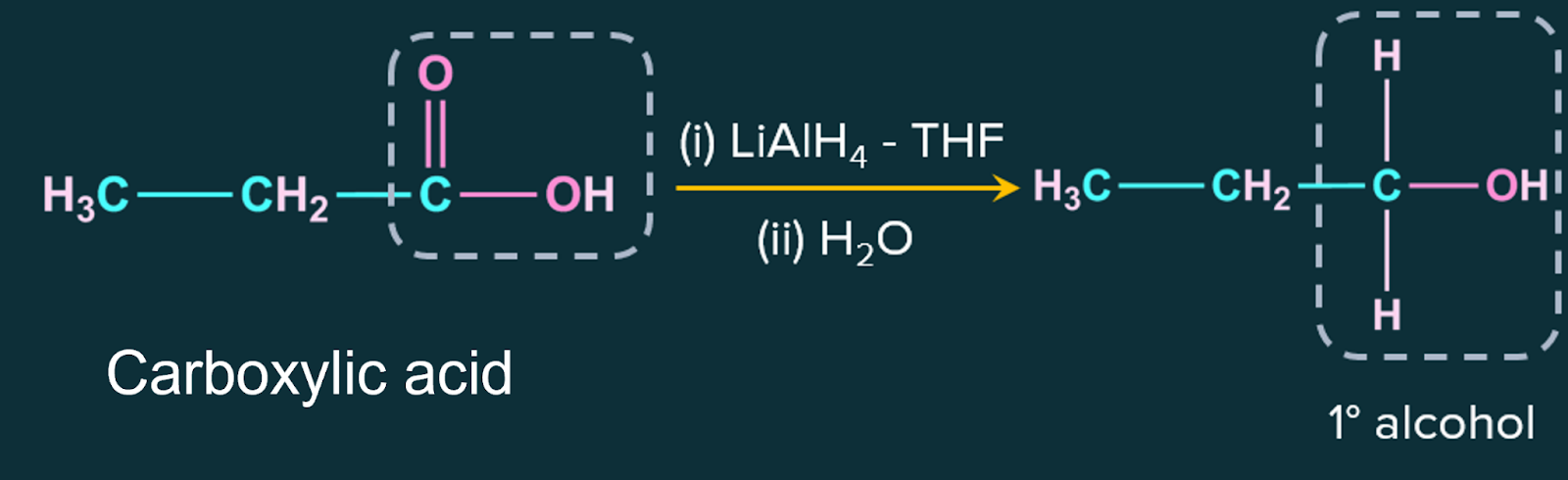
5. From carboxylic acids:
Carboxylic acids are reduced to primary alcohols in the presence of a powerful reducing agent, lithium aluminium hydride.
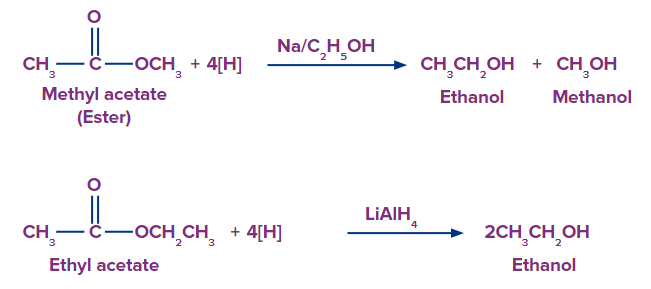
6. From esters:
When esters are converted to alcohols, two alcohols are produced: one from the acyl group and the other from the alkoxy group.
7. Preparation from Grignard reagent:
Grignard's reagents are alkyl or aryl magnesium halides that react with aldehyde, ketones, and esters. Grignard's reagents, which are based on the C–Mg link, are a very flexible reagent in organic synthesis. Grignard reagent reacts with aldehydes and ketones to produce a new product followed by acidic hydrolysis to yield primary, secondary, and tertiary alcohols. Formaldehyde produces primary alcohols, while all other aldehydes produce secondary alcohols, and ketones produce tertiary alcohols.
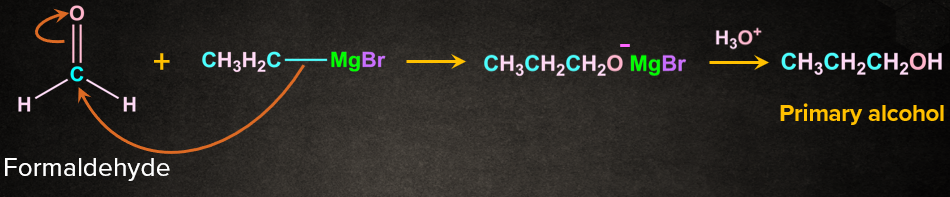
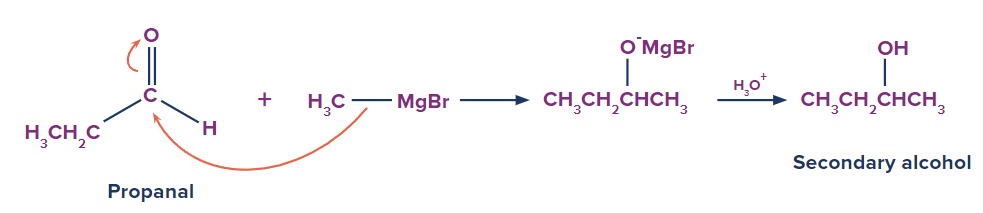
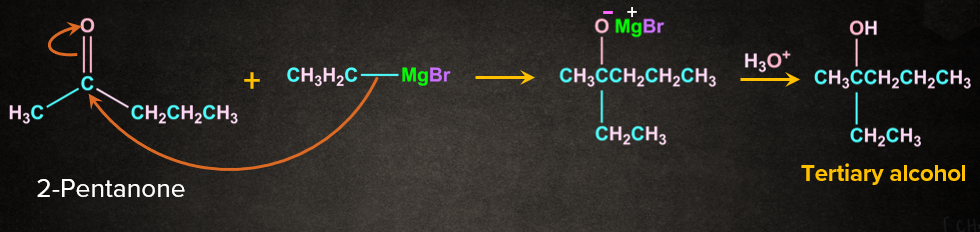
Secondary and tertiary alcohols are produced via ester.
By treating esters with Grignard's reagent, they can be transformed to alcohols. Secondary alcohols can be made by adding a suitable Grignard’s reagent to a formic acid ester and then hydrolyzing with acid.
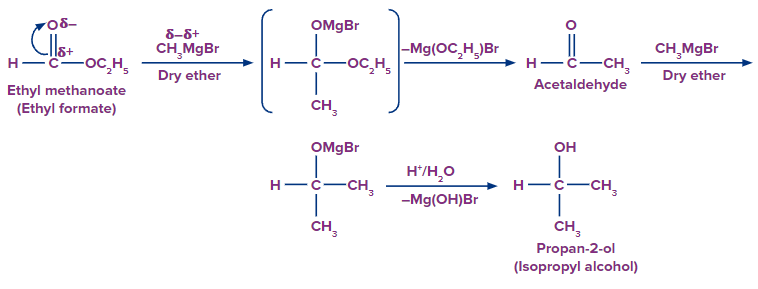
Tertiary alcohols can be produced by reaction of grignard reagent with an ester other than formic ester and hydrolyzing with the acid.
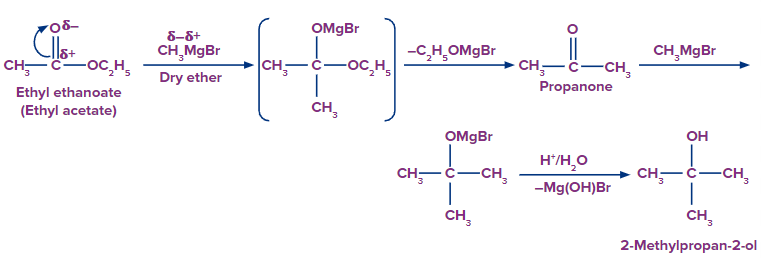
8. By reaction of ethylene oxide or oxirane with grignard reagent:
Oxy alkanes, such as oxiranes or ethylene oxide, react with a Grignard reagent in the presence of anhydrous ether to form additional products, which are hydrolyzed by water in the presence of acid to form alcohols. It's an excellent method to convert an ethylene oxide into primary alcohol.
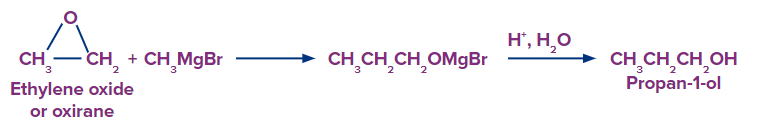
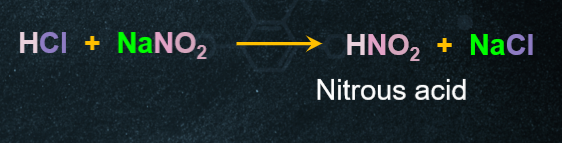
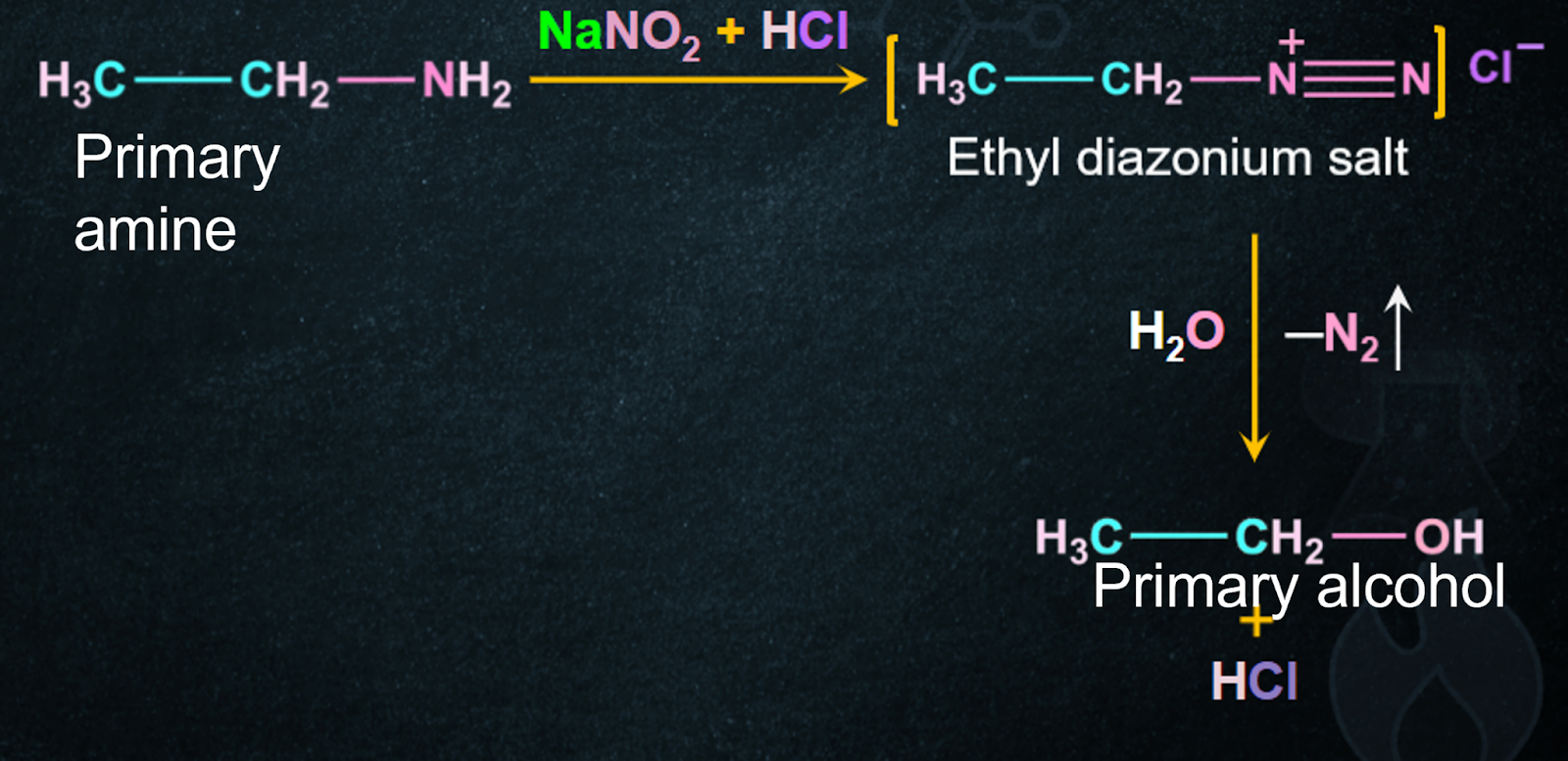
9. Preparation of Alcohol from Aliphatic Primary Amines:
When primary amines react with nitrous acid (NaNO2+HCl), alcohol is formed along with the evolution of nitrogen gas.
Industrial Preparations of Alcohols:
1. By hydration of alkenes: Alkenes are produced when petroleum is cracked. They are easily transformed to alcohol by adding water in the presence of sulfuric acid.
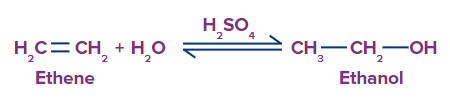
In the case of unsymmetrical alkenes, the addition takes place according to Markownikov’s rule.

2. Fermentation of molasses:
Ethanol is manufactured on an industrial scale by fermenting molasses. When sugarcane juice crystallises, the mother liquor left over is called molasses, and it has around 60% fermentable sugar. These steps are as follows:
(a) Molasses dilution: Molasses is first diluted in a 1:5 (molasses: water) volume ratio with water.
(b) Ammonium sulphate fortification: Ammonium sulphate fortification supplies yeast with an appropriate source of nitrogen.
(c) Sulphuric acid addition: A little amount of sulphuric acid is added to the fortified molasses solution. This promotes yeast development while inhibiting the growth of harmful bacteria.
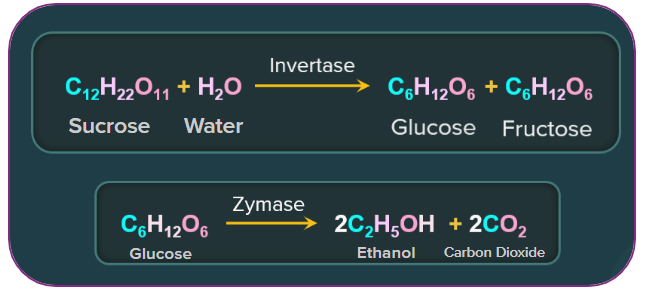
(d) Fractional distillation: The alcohol obtained during fermentation is known as "wash," and it is around 15% to 18% pure. It is turned into 92 percent pure alcohol known as rectified spirit or commercial alcohol via fractional distillation.
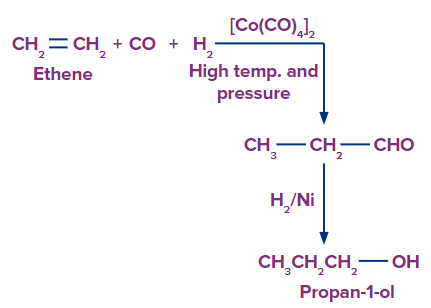
3. By Oxo process:
Aldehydes are generated when an alkene combines with carbon monoxide and hydrogen at high temperatures and pressures in the presence of an octacarbonyl dicobalt catalyst. Catalytic hydrogenation of aldehyde produces primary alcohols.
Practice problems:
Q.1. RCOOH → RCH2OH . This cannot be done by
(A) NaBH4
(B) Na/C2H5OH
(C) LiAlH4
(D) All of these
Answer: (A)
Solution: Because Na+ has a large size and cannot easily polarise the BH4- ion, NaBH4 is a weaker reducing agent. And Li+ has a high polarising power, Hence, LiAlH4 is a better reducing agent. As a result, NaBH4 is unable to reduce carboxylic acids.
Q.2. Proof spirit contains
(A) 48 % alcohol by mass
(B) 10 % alcohol by mass
(C) 5 % alcohol by mass
(D) 90 % alcohol by mass
Answer: (A)
Solution: Proof spirits contain 48.24 % alcohol by weight or 57.06 % alcohol by volume. Other spirits are labelled as overproof or underproof, with the percentage of variance indicated. A proof spirit (100 proof) in the United States is one that contains 50 % alcohol by volume.
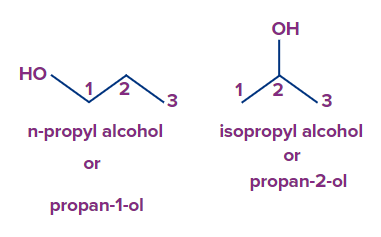
Q.3. n-propyl alcohol and isopropyl alcohol are
(A) Positional isomers
(B) Chain isomers
(C) Functional isomers
(D) none of the above
Answer: (A)
Solution: Position isomers are constitutional isomers with the same carbon skeleton and functional groups but different position of functional group. The -OH group is present on the first C atom in n-propyl alcohol. The -OH group is present on the second C atom in isopropyl alcohol.
Q.4. Ethyl alcohol is denatured by
(A) Methanol and formic acid
(B) KCN
(C) CH3OH and C6H6
(D) CH3OH and pyridine
Answer: (D)
Solution: Ethanol is naturally produced when yeast and other microbes ferment plant sugar found in plants. Denatured alcohol is ethanol that has been tainted with substances that render it unfit for human consumption. When methanol and pyridine are combined with ethanol, the result is a toxic, foul-smelling, bad-tasting, and nauseating alcohol solution. The most prevalent alcohol denaturant is methanol.
Frequently asked questions(FAQs):
What is the difference between denatured and absolute ethanol?
Answer: Absolute ethanol is hygroscopic (it attracts water), so don't expect it to last long if left uncapped. Denatured ethanol (95 % or absolute) contains additives (such as methanol and isopropanol) that make it unsafe to drink and, as a result, is exempt from certain beverage taxes.
Can you consume methylated spirits?
Answer: Methylated spirits are 70-99 % ethanol with bittering agents added to make them extremely unpleasant to drink. Because of its high ethanol content, it is poisonous by nature, and even a small drink can cause headache, dizziness, gastric disorder, nausea, and central nervous system depression.
Why is ethanol used as renewable fuel?
Answer: Because the ethanol molecule contains oxygen, the engine can burn the fuel more fully, resulting in fewer emissions and hence less pollution. Because ethanol is manufactured from plants that capture the sun's energy, it is also considered a renewable fuel.
What are the differences between ethanol and methanol?
Answer:
|
Ethanol |
Methanol |
|
Ethanol is a type of alcohol that has a carbon skeleton made up of an ethyl group. |
Methanol is a type of alcohol that has a carbon skeleton made up of a methyl group. |
|
In terms of acidity, ethanol is a weak acid when compared to water. |
In terms of acidity, methanol is a strong acid when compared to water. |
|
Alcoholic beverages are primarily composed of ethanol. |
Because methanol is extremely poisonous, it should never be used in alcoholic beverages. Typically used in the production of products such as formaldehyde. |



