-
Call Now
1800-102-2727
Discovery of electron
The discovery of electrons began with an attempt to explain the discrepancy between the behavior of cathode rays under magnetic and electric forces.
J.J Thomson in 1897 discovered the electron and determined the charge to mass ratio. In 1906, he won the Nobel prize for revolutionizing the knowledge of atomic structure by his discovery of the electron. Discovery of electrons stands out as the starting point for the research into the structure of the atom that has dominated 20th-century science.
Table of contents
- Discovery of electron
- What is an electron?
- Cathode rays
- Experimental setup used for discovery of electron
- Observations
- Conclusion
- Example
- Test your knowledge on Discovery of Electron-Quiz
- Frequently Asked Questions-FAQs
- Related topic links
What is an electron ?
Electrons are fundamental stable subatomic particles. It has a negative charge equal to -1.6 10-19 C.The mass of an electron is 9.1 10-31 kg i.e., 11837 times the mass of a proton (mass of a proton is 1.67 10-27 kg).
Experimental setup used for discovery of electron
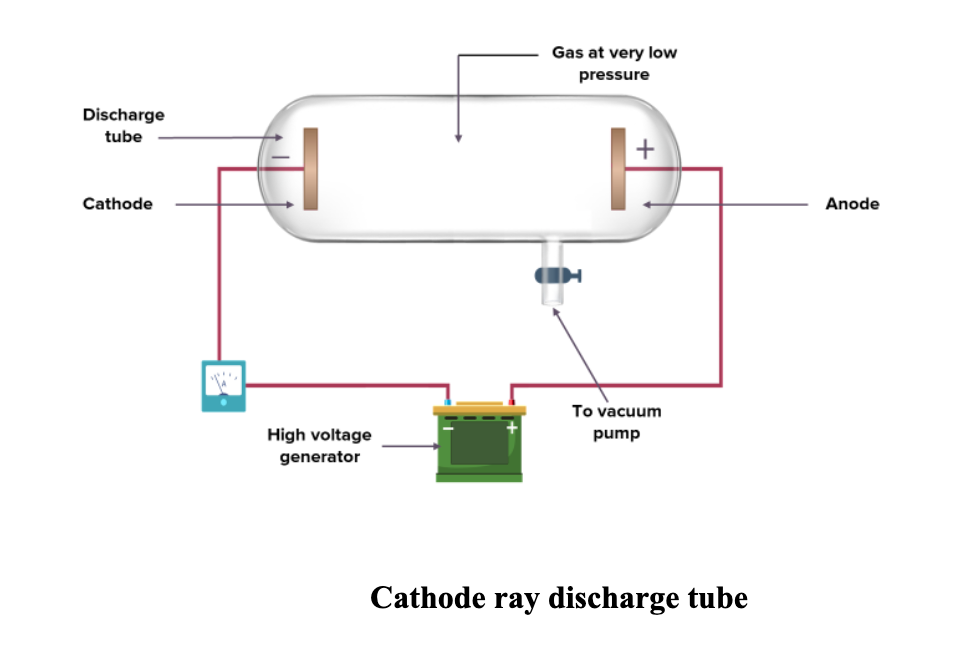
Cathode ray discharge tube
Cathode ray discharge tube: It is a cylindrical hard glass tube that is fitted with two metallic electrodes (Anode and Cathode) connected to a battery. The gas taken in the discharge tube was subjected to a very low pressure (~0.0001 atm) maintained by a vacuum pump and high voltage (~10,000 volts) to generate the cathode rays.
Cathode ray: Cathode ray is a stream of electrons observed in a discharge tube. Cathode rays are so named because they are emitted by the negative electrode (cathode) in a discharge tube.
Observations
- Cathode rays move from cathode to anode.
- Cathode rays travel in a straight line in the absence of electric and magnetic fields.
- Cathode rays are observed with the help of fluorescent screens.
- Cathode rays get deflected when placed in an electric field and magnetic field. The direction of deflection shows that they are negatively charged.
- Cathode rays rotate a light paddle wheel placed in its path.
- Cathode rays cast the shadow of an object on the ZnS screen placed in its path.
Conclusion
The above observations led to the conclusion that:
- Cathode rays consist of negatively charged particles.
- These charged particles were identified as corpuscles, later renamed as electrons.
- Cathode rays have identical properties regardless of elements used to produce them.
- Electrons are the fundamental particles. Electrons are in fact the basic constituent of all the atoms.
- Thomson disproved Dalton’s atomic theory, because according to Dalton’s atomic theory atoms were the smallest unit of the matter and they were indivisible. So, the presence of electrons fails this statement of Dalton’s atomic theory.
Recommended video: Discovery of electrons [start to 4:00 Minute)
Example 1:
From the cathode ray discharge tube experiment, it was concluded that:
(A) mass of an electron is fractional.
(B) matter contains electrons.
(C) matter contains the nucleus.
(D) None of the above
Answer: (B)
Solution: Cathode rays consist of negatively charged particles. These charged particles were identified as corpuscles, later renamed as electrons and electrons are in fact the basic constituent of all the atoms.
Example 2:
Which of the following statements is true for cathode rays?
(A) It is not deflected by a magnetic field.
(B) It does not travel in a straight line.
(C) It is not deflected by an electric field.
(D) It consists of only negative particles.
Answer: (D)
Solution: In the presence of electric and magnetic fields, the direction of deflection shows that a cathode ray is a stream of negatively charged particles called electrons.
Test your knowledge on Discovery of electron-Quiz
1. Electrons are _____ charged.
(A) positively
(B) negatively
(C) None of these
Answer: (B)
- Cathode rays travel in_______ from cathode to anode, in absence of electric or magnetic field.
(A) parabolic path
(B) circular path
(C) straight line
(D) all of these
Answer: (C)
- The cathode rays experiment concludes that:
(A) neutrons are neutral in nature.
(B) cathode ray is a stream of negatively charged particles.
(C) cathode ray is a stream of positively charged particles.
(D) All of these
Answer: (B)
- Dalton thought that atoms were the smallest units of matter - tiny, hard spheres that could not be broken down any further. Atom composed of even smaller particles was first confirmed by:
(A) Ernest Rutherford
(B) Michael Faraday
(C) J.J Thomson
(D) Antoine Lavoisier
Answer: (C)
- We know gasses are bad conductors of electricity. In cathode ray discharge tube experiment their conductivity increased by:
(A) increasing the pressure as well as the potential difference between the electrodes.
(B) decreasing the pressure as well as the potential difference between the electrodes.
(C) decreasing the pressure and increasing the potential difference between the electrodes.
(D) increasing the pressure and decreasing the potential difference between the electrodes.
Answer: (C)
Frequently Asked Questions-FAQs
- Was Thomson the first person who performed a discharge tube experiment?
Answer: No, many people performed this experiment and observed the same rays, but they were not able to conclude the property of observed rays, few famous names were Perrin and Hertz. Hertz also applied electric field to the discharge tube experiment but due to some experimental error he was not able to observe any deflection. Some researchers say Hertz experimental failure gives an idea to Thomson. Thomson introduced some quantity of air between the electrodes in the discharge tube and performed this experiment.
- Why is low pressure of gas required in cathode ray discharge tube experiment?
Answer: At low pressure, the amount of gas within the discharge tube is less thereby electrons of the cathode ray will experience very less number of collisions with the ionized gas particles. This will help electrons gain sufficient kinetic energy required to reach the anode. At high pressure, there are more gas molecules that would act as obstructions in the path of electrons, thereby preventing the electrons from reaching the anode.
- How are cathode rays produced?
Answer: Cathode rays originate from the cathode, because cathode is a negatively charged electrode. These rays strike and ionize the gas inside the discharge tube and the electrons ejected from the ionized gas travel to the anode electrode. These rays are the stream of electrons that are actually produced from the ionized gas inside the tube.
Related Topic Links:
|
Millikan’s oil drop experiment |
Discovery of protons |
|
Dalton atomic theory |
Electromagnetic radiation |
|
De broglie equation |
Thomson's atomic model |
|
Quantum numbers |
Rutherford's model of an atom |
Talk to our expert


