-
Call Now
1800-102-2727
Boyle’s Law- Statement, Equation, Representation, Applications, Practice Problems & FAQs
To understand Boyle’s law, let's look at the following setup. What do you think would happen if we press the plunger of the syringe? Yes, you got it correct! The volume will decrease but at the same time, the pressure of the air inside the syringe will increase. In the year 1662, Robert Boyle showed the relationship between pressure and volume of gas through different experimental observations and named it Boyle’s law which helps to explain different observations in everyday life.

Table of content
- Statement of Boyle’s law
- Equation of Boyle’s law
- Graphical representation of Boyle’s law
- Applications of Boyle’s law
- Practice problems
- Frequently asked questions-FAQs
Statement of Boyle’s law
According to Boyle’s law “At constant temperature and for a fixed amount of gas, the pressure of the gas varies inversely with the volume of the gas”.
Equation of Boyle’s law
Mathematically it can be represented as;
P 1V
⇒P=k1V
PV=k= Constant
Where 'P' represents pressure exerted by the gas, 'V' is the volume occupied by the gas and 'k' represents the constant of proportionality.
Note: The above relation of pressure and volume is shown considering the temperature and amount of gas to be constant.
Mathematical representation of Boyle’s law under two different pressure and volume conditions:
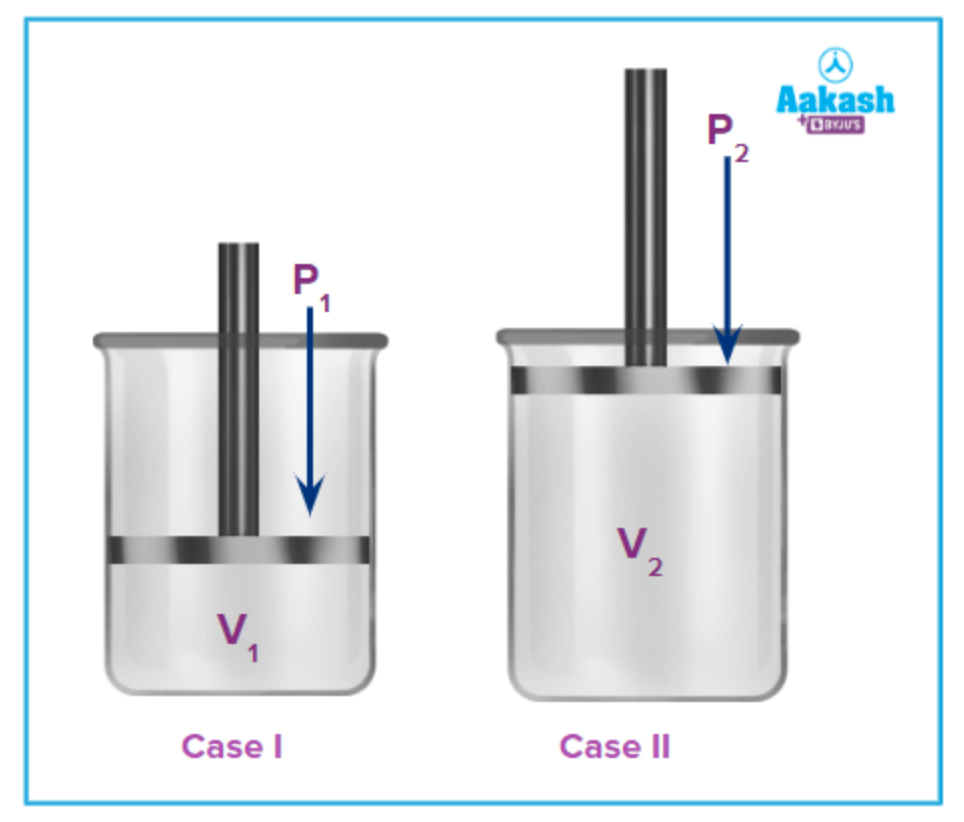
Using Boyle’s law for both Case-I and Case-II, we get;
P1V1=k ....(i)
P2V2 = k ....(ii)
From equation (i) and (ii), we get,
P1V1=P2V2=k=Constant
Where P1, V1 represent pressure exerted by the gas and volume occupied by the gas in Case-I respectively and P2, V2 represent the pressure exerted by the gas and volume occupied by the gas in Case-II respectively.
Graphical representation of Boyle’s law
The P-V curve for a fixed amount of gas and at a constant temperature is represented as;
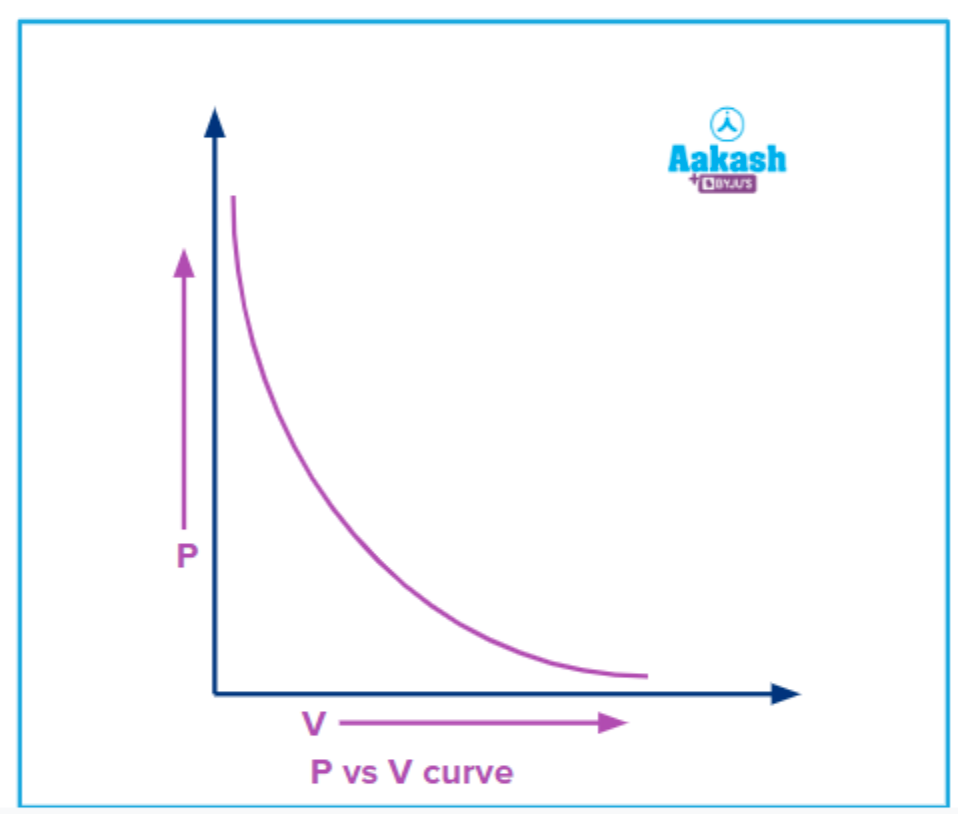
According to the equation;
P 1V
Pressure exerted by the gas is inversely proportional to the volume occupied by the gas, so if pressure (P)is taken on the Y-axis and volume (V) on the X-axis, the nature of the curve would be rectangular hyperbola and you can see from the graph that pressure is increasing when the volume is decreased.
Applications of Boyle’s law
- Filling of natural gas in containers
- Working of a cylinder-piston arrangement
- Wearing spacesuit in space
- Ascending gradually during scuba diving
Practice problems
1. A 1.03 L cylinder with flexible walls containing nitrogen at a pressure of 710.6 torr is allowed to expand at constant temperature such that the final pressure of the nitrogen becomes 583.1 torr.The final volume of the cylinder is: (Assuming nitrogen present in the flask behaves ideally)
a. 1.255 L
b. 1.34 L
c. 0.225 L
d. 0.14 L
Answer: (A)
Solution:
According to the given data,
Initial volume of the cylinder (V1)=1.03 L
Initial pressure of nitrogen gas in the cylinder (P1)=710.6 torr
Final pressure of nitrogen gas in the cylinder (P2)=583.1 torr
Let the final volume of the cylinder be V2(L)
Using Boyle’s law we get,
P1V1=P2V2
Putting the values in the above expression we get,
710.6 torr1.03 L=583.1 torrV2
V2=1.255 L
2. An ideal gas undergoes compression at a constant temperature, which reduces its volume by2.20 dm3. The final pressure and the volume of a gas are 5.04 bar and 4.65 dm3 respectively. Calculate the original pressure of the gas in (bar).
Solution: Let the original pressure be P1.
Initial volume of the gas (V1) = Final volume of the gas (V2) + Reduction in volume
=4.65 dm3+2.20 dm3
V1=6.85 dm3
Final pressure exerted by the gas (P2)=5.04 bar
Using Boyle’s Law and putting the values we get,
P1V1=P2V2
P16.85 dm3=5.04 bar4.65 dm3
P1= 3.42 bar
Q3. Calculate the percentage change in volume of an ideal gas if it is compressed at 55C from1.00 atm to 50.00 atm.
Solution: Let the initial and final volume of an ideal gas be V1(L) and V2(L) respectively.
Initial pressure (P1)=1 atm
Final pressure (P2)=50 atm
Using Boyle’s Law and putting the values we get,
P1V1=P2V2
1 atmV1=50 atmV2
⇒ V2V1=150
% Change in volume = V2-V1V1100 %
=(V2-V1V1100 %)
={(V2V1-1)100}
Putting value of V2V1 from equation (i);
={(150-1)100}
=-98%
Note: -ve sign here signifies a decrease in volume of an ideal gas.
Q4. Slope of the plot between PV and V at constant temperature is_______.
a. 1
b. 12
c. 12
d. 0
Answer : (A)
Solution:
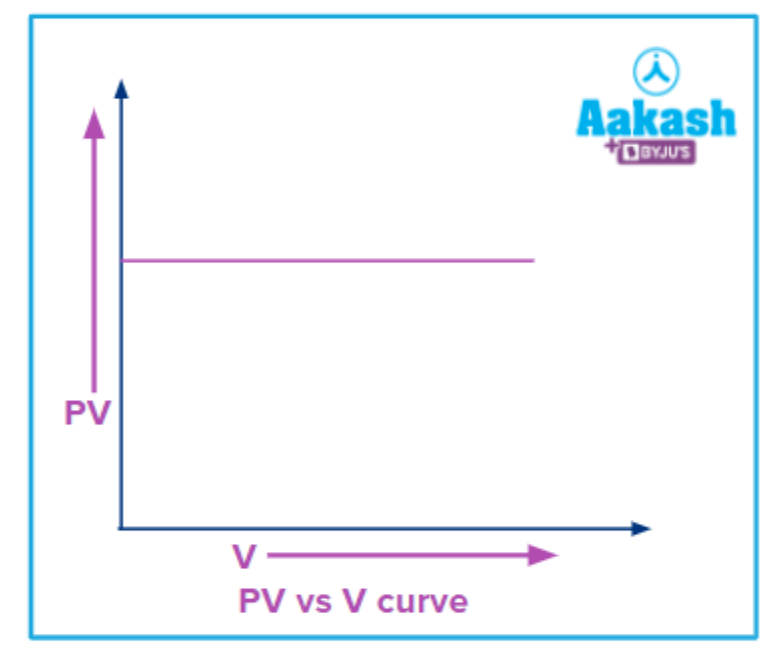
As shown in the graph above plot is parallel to the X-axis and therefore,
Slope =tanθ=tan 0°=0
Frequently asked questions-FAQs
1. How does Boyle's law have practical application in scuba diving?
Answer: We are aware of the fact that according to Boyle's law, pressure and volume are inversely related to each other. So, if a diver takes a lung full of air underwater, that air will expand his lungs as he rises to the surface because the pressure is higher underwater and at the surface, pressure is reduced. If he ascends too rapidly or holds his breath the expanding air can rupture his lungs.
2. How density of gas and pressure can be related considering the gas obeying Boyle’s law?
Answer: According to Boyle’s law we know that pressure and volume are inversely related considering temperature and amount of gas to be constant.
Density =mass of the gasvolume occupied by the gas, since the amount of gas is assumed to be constant.
Therefore, Density of the gas 1volume occupied by the gas....(i) .
According to Boyle’s law,
Pressure of the gas 1Volume of the gas....(ii)
So, considering (i) & (ii), it can be said as;
Density of the gas is directly proportional to the pressure of the gas.
Q3. Why do inflated balloons burst when it is compressed?
Answer: When the inflated balloon is compressed its volume decreases. We know, according to Boyle’s law, pressure is inversely proportional to the volume of the gas. Therefore, on decreasing the volume of the inflated balloon pressure inside the balloon increases and it burst.
Q4. Why are gases highly pressurized while storing them in a small container?
Answer: Gases are generally pressurized when stored in a small container. We know, according to Boyle’s law, pressure is inversely proportional to the volume of the gas. Therefore, pressurizing the gas decreases the volume occupied by the gas and a larger volume can be stored in the small containers.
Related topics
|
Charles's Law |
Gay-Lussac’s Law |
|
Avogadro’s Law |
Dalton's Law of Partial Pressure |
|
Ideal Gas Equation |
Real Gas |


