-
Call Now
1800-102-2727
Uses of Aldehydes: Introduction, Properties of Aldehydes, Uses of Aldehydes, Practice Problems & Frequently Asked Questions
Are you a favourite of nail polish? Have you ever wondered what nail paints are, its bright, clear, and provide a lovely image when used.
These polishes have a silky feel and an appealing appearance because of a chemical compound namely acrolein, that is included in them. Additionally, acrolein used to make glue.
Everybody must have tasted vanilla ice cream and for some, it may be the tastiest ice cream. But, do you know that an aldehyde is added to give vanilla flavour to the ice cream?
An aromatic aldehyde known as vanillin, which is technically known as 4-hydroxy-3-methoxybenzaldehyde, is the flavouring ingredient in vanilla ice cream.
Aldehydes are mostly employed as flavouring, solvent, and fragrance components as well as intermediates in the production of dyes, polymers, and medicines. The flavouring ingredients naturally include certain aldehydes. Benzaldehyde, which gives fresh almonds their flavour and aroma, cinnamaldehyde, oil of cinnamon, and vanillin, the primary flavouring in vanilla beans, are a few examples.
Table of Contents:
- Introduction of Aldehydes
- Properties of Aldehydes
- Uses of Aldehydes
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Aldehydes:
Aldehydes have a carbonyl group (>C=O) with one side attached to a hydrogen atom. A second hydrogen atom or a hydrocarbon group like an alkyl or aryl group can be attached to the carbonyl group's other side. R stands for the alkyl or aryl group, and the basic structural formula for aldehydes is
(R-CHO). The carbonyl carbon of the aldehyde group has a sp2 hybrid orbital. A triangular planar structure can be seen in the aldehyde. The bond angle of this structure is about 120o.
Due to the polarisation of the carbonyl group caused by carbon-oxygen electronegativity differences, aldehydes are nucleophilic at the oxygen atom and electrophilic at the carbon atom (oxygen is more electron-withdrawing than carbon).

Properties of Aldehydes:
Physical State of aldehydes:
Apart from formaldehyde, which is a gas at ambient temperature, the most prevalent aldehydes and ketones are liquids at normal temperatures. Higher molecular weight aldehydes and ketones have pleasant aromas, whereas lower molecular weight aldehydes have strong and disagreeable aromas. Numerous ketone compounds are employed in the production of perfumes, and some aromatic aldehydes obtained from natural sources have a highly attractive fragrance.
Dipole moment in aldehydes:
The carbonyl group of aldehydes has a double bond between carbon and oxygen. Additionally, the shared electron pairs are drawn by oxygen since it has a stronger electronegative charge than carbon, which increases the carbonyl group's reactivity and polarity. Aldehydes thus have large dipole moments.
Aldehydes contain a double bond between carbon and oxygen in the carbonyl group. Additionally, oxygen draws the shared pairs of electrons since it has a higher electronegative charge than carbon, increasing the reactivity and polarity of the carbonyl group. Thus, aldehydes have significant dipole moments.
Solubility in water:
Smaller aldehydes are readily miscible with water, but their solubility decreases as chain length increases. For example, the common minor aldehydes methanal and ethanal are miscible with water in all quantities. The solubility is due to the fact that, whereas aldehydes cannot create hydrogen bonds with one another, they can establish hydrogen bonds with water molecules. A hydrogen bond can be established when one of the slightly positive hydrogen atoms in a water molecule is strongly attracted to one of the lone pairs on an aldehyde's oxygen atom.
Of course, there will also be dipole-dipole interaction between the water molecules and the aldehyde as well as dispersion forces. Energy is released during the formation of these attractions, which aids in supplying the energy required to isolate the molecules of water and aldehyde molecules from one another before they can mix.
The hydrocarbon "tails" of the molecules—all the hydrocarbon parts outside the carbonyl group—begin to obstruct movement as chain lengths grow. They break the relatively strong hydrogen bonds that exist between water molecules by compressing them and preventing anything better from doing so. Solubility declines as a result of the process being less energetically profitable.
Boiling Point of aldehydes:
Aldehydes have higher boiling temperatures than other non-polar compounds due to the presence of dipole-dipole interactions between carbonyl compound molecules, which are substantially stronger than van der Waals forces between alkane molecules. Aldehydes have lower boiling points than similar amounts of carboxylic acids and alcohols, this is because they do not establish hydrogen bonds with one another.
The boiling point of methanal, a gas, is 252 K, while the boiling point of ethanal is 294 K. Therefore, ethanal boils at temperatures that are nearly normal. The boiling temperatures of the other aldehydes which are both liquids, increase with the size of the molecules. The magnitude of the intermolecular forces determines the size of the boiling point.
Uses of Aldehydes:
Formaldehyde:
Although formaldehyde is primarily produced by the oxidation of methanol, it is often sold as a 37% solution in water known as formalin. Although it can be used as a fungicide, germicide, and pesticide, as well as for the preservation, tanning, and its most prevalent application is in the production of some polymeric materials.
Mortuaries and medical labs frequently employ formaldehyde as preservation and industrial disinfectant. By causing molecular changes in the tissue, formaldehyde stops germs from eating it. It deconstructs the structures of your tissue.
Urea-Formaldehyde:
Urea-formaldehyde, commonly known as urea-methanal, is a non-transparent thermosetting synthetic resin or polymer. It is composed of urea and formaldehyde. These resins are used in glues, varnishes, plasterboard and moulded items.
Phenol-Formaldehyde:
A reaction between phenol and formaldehyde can create the material Bakelite. It has a three-dimensional structure instead of a linear chain. A wide range of goods, including bangles, car parts, and cutlery handles, are made from bakelite.
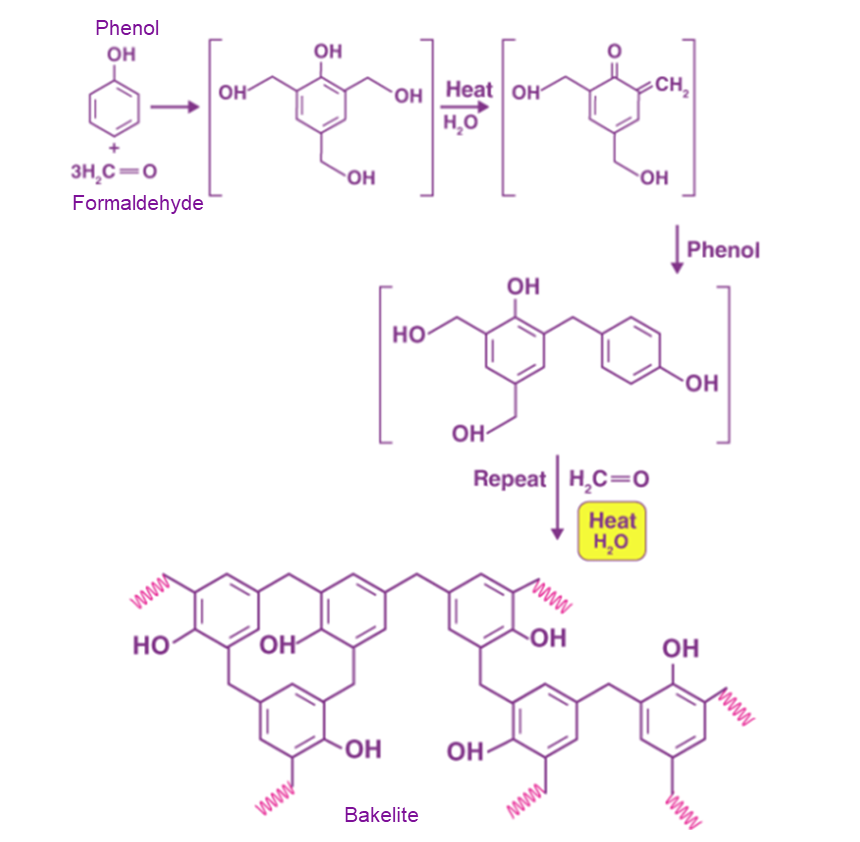
It can be coloured in a variety of ways, enabling the production of colourful goods that are pleasing to the eye. In order to create non-conducting components for radios and electric equipment, such as plugs, wire insulation, switches, and automotive distribution caps, bakelite is utilised as a good insulator. Watches, keys, washing machines, dolls, cookware, and other goods are made using it.
Aldehyde-containing compounds:
Carbohydrates (such as cellulose, starch and sugars), which are based on substances with an aldehyde group in addition to the hydroxyl groups, are examples of aldehydes. Another example is retinal, an aldehyde that, upon incorporating with a protein (or opsin) in the eye's retina forms rhodopsin, the main compound involved in the vision process.
Rhodopsin experiences cis-trans isomerization in the retina in response to light exposure. A nerve impulse that is produced as a result of the altered molecular shape may be what causes the brain to receive the nerve impulse and interpret it as a visual signal.
A crucial role for aldehydes is played in the caramelization of sugars. The amino acids transform the sugar's aldehyde group into an unsaturated aldehyde during slow cooking. Sugar acquires the distinctive caramel-brown colour as a result.
In Indian cuisine, certain spices, like coriander, are crucial. Coriander's distinctive aroma is caused by the presence of specific aldehydes. Coriander is made up of approximately 40 various chemical compounds, with 82 % of them being aldehydes with 9-10 carbon atoms, which are solely accountable for the smell of coriander leaves.
Aldehydes are a necessity for numerous industrial operations, including making adhesive, tanning leather, and creating polymeric products.
Aldehydes like glutaraldehyde function as fungicides, disinfectants, and insecticides as well. It interferes with the activity of bacteria and fungi by attacking their cell walls and membranes. Additionally, formaldehyde is employed in drug screening and photographs.
Acrylic acid, which is used to manufacture plastics, glue, floor polish, and paints, is made from acrolein. Hyacinth and other plants frequently choke streams, making navigation challenging. To clear the water and stop the growth of these plants, acrolein is applied.
Acetaldehyde is primarily used to create acetic acid, but it is also used to create ethyl acetate, peracetic acid, derivatives of the pyridine ring, perfumes, colours, polymers, and artificial flavourings.
In Perfumes:
There are very few fragrances in the world that do not contain aldehydes due to their symbiotic relationship with the perfume industry.
These chemical components are what give many natural products, like rose, vanilla, orange peel, pine essence, citronella, and cinnamon essential oils, their pleasant aromas.
They can also be artificially created in a lab.
The formyl group, which is a component of a molecule with a certain structure (R-CHO) in organic chemistry, is present in these fragrant amalgams.
Although aromatic aldehydes have extremely complex chemical structures, their fragrance makes them easy to recognise.
An amalgam containing the -CHO radical is referred to as an aromatic aldehyde, such as the almond-like smelling benzaldehyde. These chemical elements typically give a perfume formula a soapy-waxy-lemony-floral touch.
Practice Problems:
Q1. Which kind of hybridization does the carbonyl carbon in aldehydes exhibit?
(A) sp3
(B) sp2
(C) sp
(D) None of the above
Answer: (B)
Solution: Aldehydes are compounds that include one hydrogen atom linked to an alkyl or aryl group. Ketones only have aryl or alkyl groups attached to the carbonyl carbon. Thus the hybridization of the carbonyl carbon in both aldehydes and ketones is sp2.
Q2. Aldehydes and ketones are
(A) Functional isomers
(B) Chain isomers
(C) Position isomers
(D) None of the above
Answer: (A)
Solution: Ketones and aldehydes are functional group isomers. Both aldehydes and ketones contain the carbonyl group, or C=O. The functional group is connected to one or two hydrogen atoms in the case of aldehydes and to two carbon atoms in the case of ketones. For instance, propanal and propanone are functional isomers because they have the same chemical formula (C3H6O), but distinct functional groups
(-CHO and R(CO)R, respectively).
Q3. In order to preserve biological specimens, which one of the following is often used?
(A) Methanal
(B) Ethanal
(C) Acetone
(D) Benzaldehyde
Answer: (A)
Solution: As formalin (40 % methanal, 8 % methanol, 52 % water) is commercially available and easily miscible in water, it is employed to preserve biological specimens.
Q4. Which one of the following is not a use of formaldehyde?
(A) Production of bakelite
(B) Biological specimen preservation
(C) manufacturing of acetic acid.
(D) None of the above
Answer: (C)
Solution: Since formaldehyde has just one carbon atom, it cannot serve as the first ingredient in the production of acetic acid. However, it is employed in the production of polymeric goods, adhesives, and bakelite.
Frequently Asked Questions(FAQs):
Q1. How do aldehydes and ketones differ?
Answer: The location of the carbonyl group inside the molecule distinguishes aldehydes and ketones. A carbonyl group is joined to a carbon atom at the end of a carbon chain to form an organic molecule known as an aldehyde. An organic molecule known as a ketone is one in which the carbonyl group is joined to a carbon atom within the carbon chain.
Q2. Even though both alkenes and carbonyl compounds have a bond, alkenes exhibit electrophilic reactions while carbonyl compounds exhibit nucleophilic addition reactions. Clarify.
Answer: Aldehydes and ketones exhibit nucleophilic addition, but alkenes exhibit electrophilic addition because the double bond in alkenes links two carbon atoms together with no polarity as a result. In contrast, a double bond links atoms with distinct polarity in carbonyl compounds. The carbonyl bond is susceptible to a nucleophile addition reaction because of its polarity.
Q3. The typical reactions of the carbonyl group are not formed by carboxylic acids. Explain?
Answer: Carboxylic acids do not exhibit the typical carbonyl group reactivity. This is due to resonance occurring between the lone pairs on the oxygen atom linked to the hydrogen atom in the -COOH group, which reduces the electrophilicity of the carbon atom. As a result, carboxylic acids do not exhibit the typical reactions of the carbonyl group.
Q4. For what reason are aldehydes more reactive than ketones?
Answer: Ketones are less reactive than aldehydes.
Two things affect the carbonyl group's reactivity:
(i) the quantity of the positive charge on the carbonyl atom (electrophilic character) and
(ii) Steric crowding on the carbon atom in a carbonyl compound (steric effect)
Aldehydes only include one alkyl group (+I effect), whereas ketones have two of these groups. Accordingly, the positive charge on the carbonyl atom in aldehydes is greater than the positive charge on the carbonyl atom in ketones. Additionally, aldehydes are less crowded than ketones.



