-
Call Now
1800-102-2727
Anomalous behaviour of boron- Definition, Important compounds, properties, Anomalous properties, Comparison, Practice problems, FAQs.
Have you noticed that the youngest kids in the family always demand toys, chocolate and many more? This may happen in your house also. Let us understand this through the story of Tintin, the smallest kid in a family of 5 members. Grandparents, mom, dad, and his elder sister, all loved Tintin deeply. He was not only the most adored and pampered one, but also he was the one with maximum tantrums!
How powerful are a family's youngest members? Every family will most likely have a unique perspective on this. However, one thing is certain: the youngest members are unquestionably the most important. This was major because he was so small, of course, because of his age !!
Hey, wait, this is definitely not a story of Tintin and his adventures! But why I really wanted to draw your attention is to the fact that the smallest of the Group 13 family, i.e., Boron, is also one such member with certain 'tantrums'. Basically, these features of 'Boron' which are quite different from its fellow family members, are what we termed as 'Anomalous properties'.

Table of content:
- What is Boron?
- Important compounds of boron
- Properties of boron
- Anomalous properties of boron
- Comparision of Aluminium and Boron
- Practice problems
- Frequently asked questions.
What is boron?
Placed next to carbon in the Periodic Table of Elements, boron is a metalloid - a substance with both metallic and nonmetallic properties. Boron belongs to Group 13, Period 2. It is one of the most common elements on the planet. It makes up less than 0.0001% of the mass of the earth's crust. It comes in a variety of forms, including borax, orthoboric acid, and kernite. Boron is found as borax in Rajasthan and Ladakh, India.
Boron is a highly reactive element, and many different boron compounds can be found in nature. These compounds have been found to be extremely useful in the production of a variety of products in a variety of fields.
Important compounds of Boron:
Some of the most important boron compounds found in nature are named below.
- Halides of Boron:
The halides of boron include BF3, BCl3, BBr3 and BI3. These are some examples of electron-deficient molecules as their octet is incomplete since the boron has six electrons in its outermost shell in the monomeric trihalide form. As the monomeric trihalides are electron-deficient, they are strong Lewis acids and form Lewis acid-base adducts.
Lewis acids are chemical compounds with vacant orbitals that can accept electron pairs from Lewis bases.
The order of Lewis acidity among boron trihalides is
BF3< BCl3 <BB3< BI3.
Example: H3N : →BF3Here, ammonia donates its lone pair of electrons to the empty p-orbital of boron. Boron is sp2 hybridised, but after accepting the lone pair of electrons from ammonia, it becomes sp3 hybridised.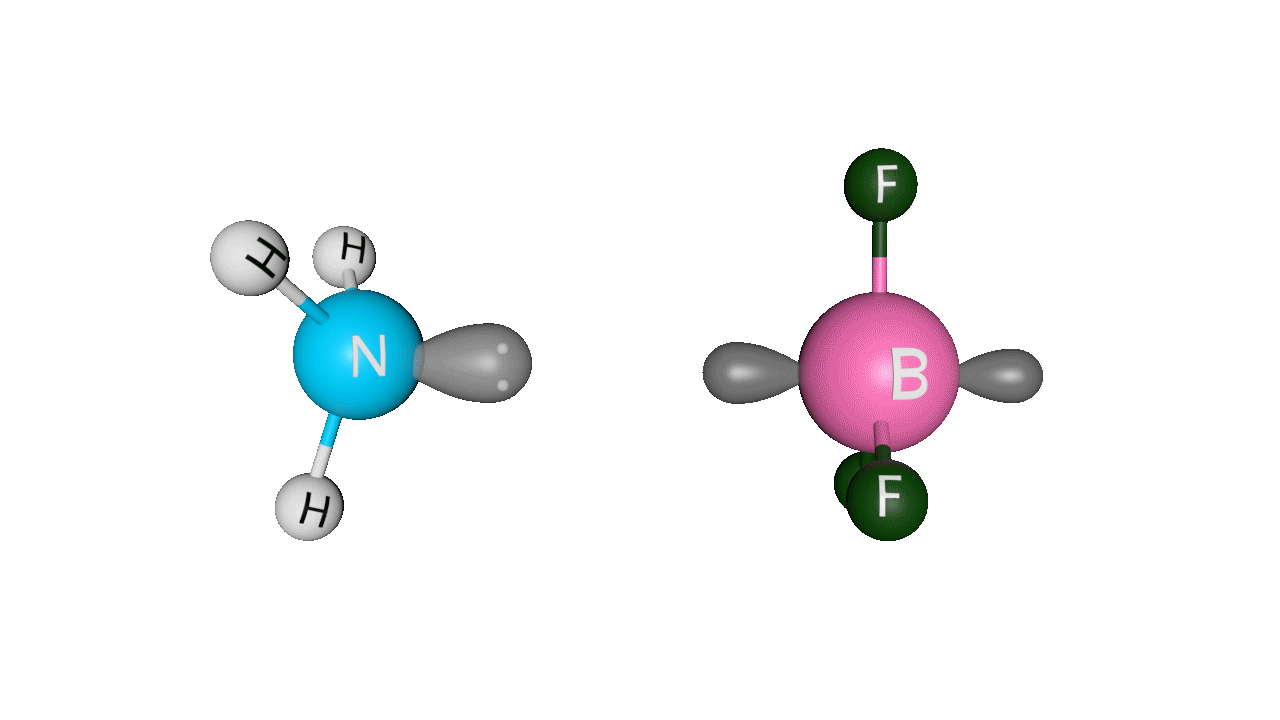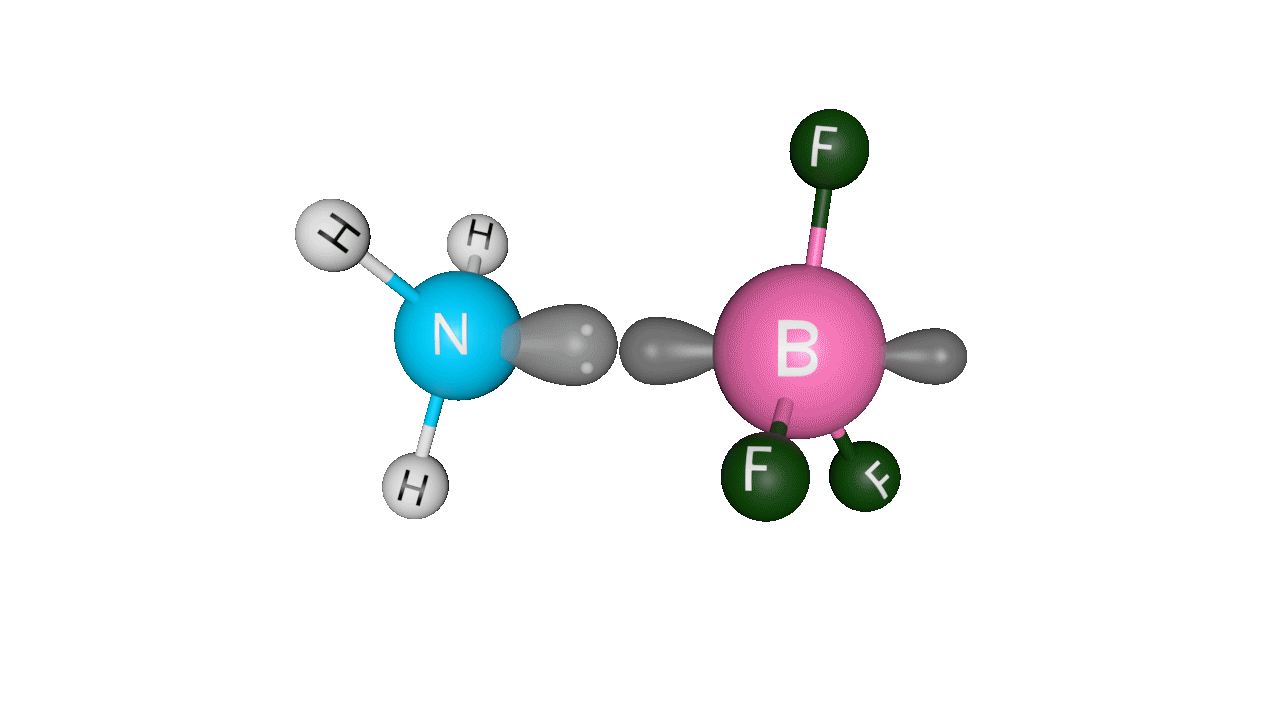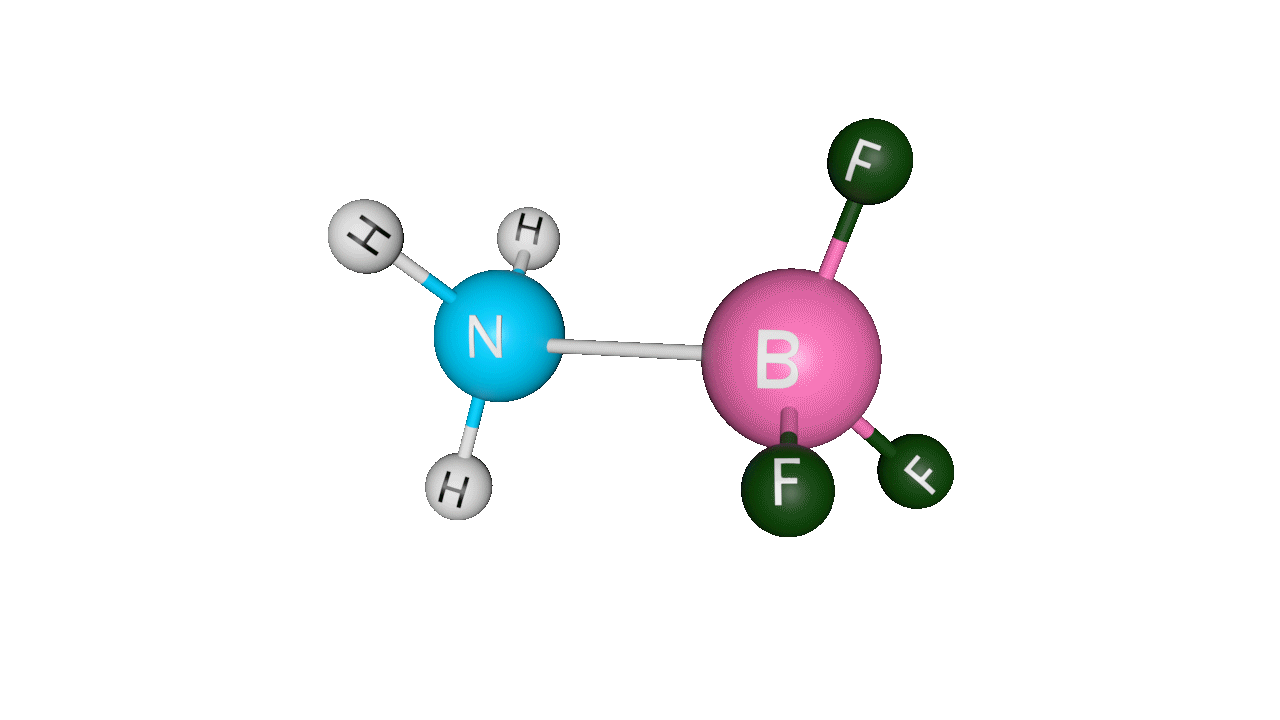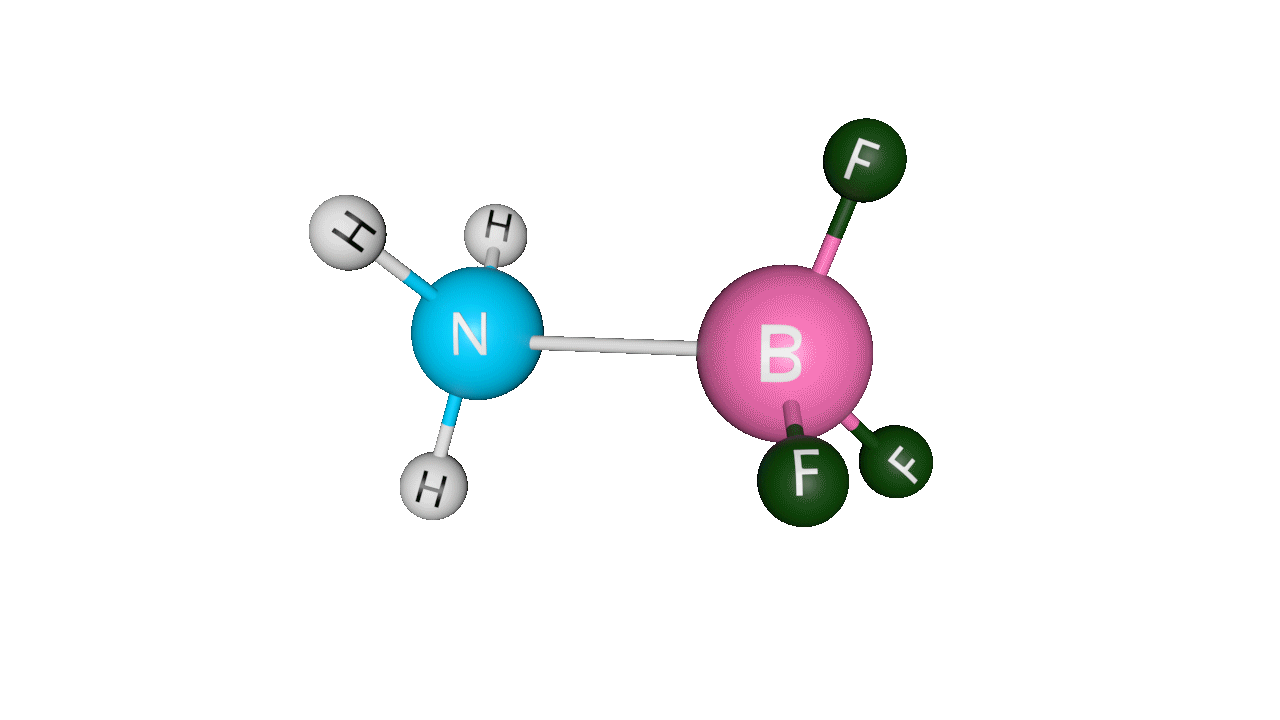
![]()
- Boron hydrides:Although none of the Group 13 elements reacts directly with hydrogen, there are several "interesting hydrides" that have been discovered. By comparison with alkanes, boron hydrides are sometimes referred to as boranes (hydrocarbons). Nearly 20 boranes have been identified, with 11 of them being well-characterized. They are divided into two groups:
- Closo boranes (BnH(n+2))
Some examples are B2H4, B5H7 etc
- Nido boranes (BnH(n+4))Some examples are
- Arachno boranes (BnH(n+6))Some examples are:
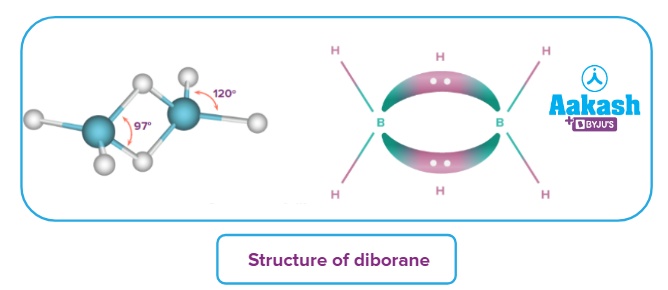
Of all the boranes, the one studied extensively is diborane. It is a dimer of borane (BH3). Following are the important points pertaining to the structure of diborane.
- The simplest boron hydride is diborane. The four-terminal hydrogen atoms and the two boron atoms lie in one plane. Above and below this plane are two bridging hydrogen atoms.
- The four-terminal B-H bonds are regular two centre-two electron bonds, while the two bridge (B-H-B) bonds (banana bond) are three centre-two electrons (3c-2e) bonds.
- The bridging bonds are longer and weaker as compared to the terminal bonds.
- The hybridisation of boron is sp3
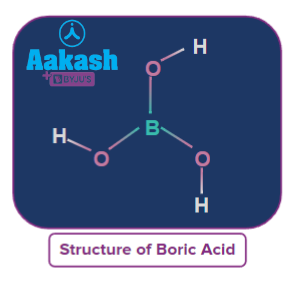
- Orthoboric Acid:It is also known as boric acid or hydrogen borate. Each boric acid molecule features boron-oxygen single bonds. The boron atom occupies the central position and is linked to three hydroxyl groups. The overall molecular geometry of boric acid is trigonal planar, and its molecular formula is H3BO3In orthoboric acid, boron has sp2 hybridisation. In orthoboric acid, oxygen has only two bonds (one with B and one with H) and two lone pairs. Therefore, oxygen should be in sp3 hybridisation, but orthoboric acid is found in the 2D sheet. Thus, it should be in sp2 hybridisation.
Orthoboric acid is a mild monobasic that is soluble in water. It accepts OH-rather than donating protons like most acids. As a result, it's a Lewis acid, and it's better expressed as B(OH)3.
Video link: https://www.youtube.com/watch?v=RObKTxtGtV8
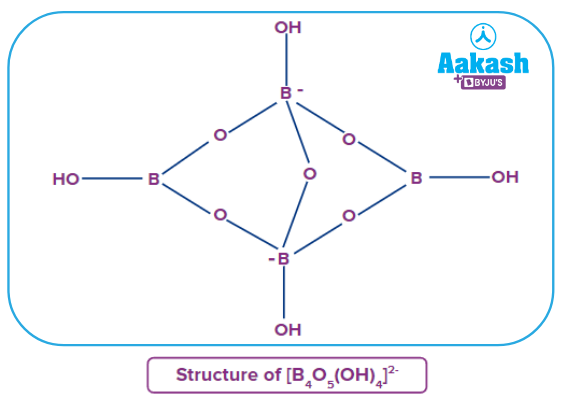
- Borax:Borax is a compound consisting of boron, united to oxygen and sodium. It contains tetranuclear units [B4O5(OH)4]2-. The formula of borax is Na2[B4O5(OH)4].8H2O. Borax dissolves in water to give an alkaline solution. It contains five B−O−B linkages.
Anomalous behaviour of Boron:
Boron deviates significantly from the rest of the members of group IlIA, as evidenced by the following characteristics:
- Because boron has such small atomic radii, the outermost electrons are more attracted to the nucleus. Its ionisation energy is really high. Because of this, boron has a nonmetallic property, whereas the rest of the elements are metals.
- Due to the lack of d-orbitals, boron has a maximum covalency of four, while the other elements have a maximum covalency of six.
- Boron is the only element that shows allotropy.
- Boron has an icosahedral shape and is a symmetrical solid.
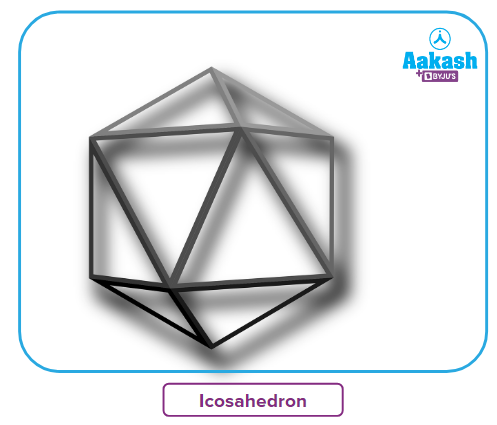
- Boron has an oxidation state of +3, while others have oxidation levels of +1 and +3.
- Because the hydration energy is smaller than the sum of three successive ionisation energies, boron does not form cations in an aqueous solution.
- Boron halides are monomeric, whereas other element halides are dimeric. There is no evidence of the inert pair effect.
- Boron does not degrade water or steam, although it does decompose hot water and steam.
- Boron's oxide, B2O3, is acidic in nature, whereas the oxides of the other elements are either amphoteric or basic.
- Non-oxidising acids do not damage boron, however, they do affect other elements.
- Boron dissolves in HNO3 condensate to generate H3BO3. The other elements, particularly Al and Ga, become inactive.
- Boron forms borides when it reacts with metals, but it does not react with other elements. They have the ability to produce alloys.
Comparison of Boron and Aluminium:
Because both boron and aluminium have three electrons in their valency shells, their properties are very similar.
|
Boron |
Atomic number: 5 |
2,3 |
||
|
Aluminium |
Atomic number: 13 |
2,8,3 |
They do, however, contain differing numbers of electrons in their penultimate shells (2 in boron and 8 in aluminium), resulting in significant differences.
Dissimilarities between Boron and Aluminium:
Because of differences in electrical arrangement, size, and ionisation potential, boron and aluminium have different characteristics. The table below lists the main points of distinction
|
S.No. |
Boron |
Aluminium |
|
1 |
Boron is a predominantly non-metallic element |
Aluminium is a predominantly metallic element. |
|
2 |
It has a high melting point (m.pt. 2300℃). |
Its melting point is low (m.pt. 660℃). |
|
3 |
It is unaffected by dil.HCl and dil. H2SO4 . Conc.HNO3 reacts with boron.B(s)+Conc.HNO3(l)H3BO3(l)+3NO2(g) |
It dissolves in HCl and dil. H2SO4 and produces hydrogen.With conc. HNO3', it becomes inactive. A thin layer of Al2O3 is produced on the metal's surface, rendering it inert. |
|
4 |
Borates have a high level of stability. |
Aluminates have lower stability. |
|
5 |
It exclusively forms covalent compounds. |
It is capable of producing both covalent and electrovalent molecules. |
|
6 |
BCl3 is a combustible liquid. |
AlCl3 is a solid substance. |
|
7 |
Boron has an allotropy property. Crystallineboron is the allotropic form. With boron that is amorphous |
Allotropy does not exist in aluminium. |
|
8 |
B(OH)3 is a weak acid. |
Al(OH)3 is amphoteric. |
Practice Problems:
Q1. Among these, which one is an incorrect statement regarding B2H6 ?
A. B2H6 is a colourless gas that is extremely poisonous.
B. B2H6 has a boiling point of 180 K.
C. B2H6exist as gases at room temperature, and to make them solid or liquid, they have to be cooled.
D. B2H6exist as solid at room temperatureAnswer: D
Solution: B2H6 is a colourless and highly toxic gas with a boiling point of 180 K. There are no strong intermolecular attractions between B2H6 molecules. Thus, they exist as gases at room temperature, and to makes them solid or liquid, they have to be cooled.
Q. 2. Boric acid forms a polymeric structure due toA. Its acidic natureB. Monobasic natureC. presence of H-bondingD. Geometry
Answer: BSolution: Boric acid forms a polymeric structure, connecting the H3BO3 molecules through hydrogen bonds. The structure of boric acid is mentioned below.
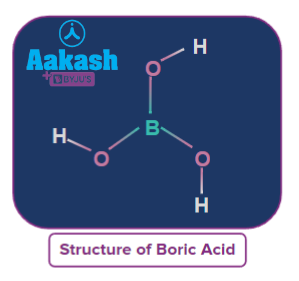
Q3. B(OH)3 (aq) + NaOH (aq) Na[B(OH)4](aq)
The addition of which of the following proceeds the reaction in the forward direction?
A. Cis-1,2-diolB. Trans-1,2-diolC. BoraxD. Na2HPO4
Answer: We know that the reaction of orthoboric acid with hydroxyl ion is a reversible reaction and cis-diol forms a very stable complex with [B(OH)4]-, removing it from the solution. Thus, the removal of one of the products shifts the equilibrium in the forward direction (according to Le-Chatelier's principle).
So, option (A) is the correct answer.
Q4. _____ is also used as a catalyst in the manufacture of polyisobutene in large quantities.
A. BCl3
B. BF3
C. H3BO3
D. B2H6
Answer: B
Solution: BF3 is also used as a catalyst in the manufacture of polyisobutene in large quantities.The supported catalysts are active in isobutylene carbocationic polymerization and may be recovered and reused for several reaction cycles without losing their activity.
Frequently asked questions-FAQ
Q1. What is the most typical location for boron to be found?
Solution: The mining of boron-containing minerals such as colemanite, ulexite, tincal, and kernite is the principal source of both boron and borates. Only a few deposits are economically viable for mining. These can be found in Turkey and the United States' dry regions, as well as Argentina, Chile, Russia, China, and Peru.
Q2. Is boron found in natural substances that humans can consume?
Solution: Boron is a trace mineral that is involved in a number of physiological processes. Dried fruit, nuts, and vegetables are the finest plant-based boron sources as well as wine!
Q3. What role does boron play in everyday life?
Solution: Boric (or boracic) acid, borax (sodium borate), and boric oxide are the most significant boron compounds. Eye drops, mild antiseptics, washing powders, and tile glazes all contain these ingredients. Borax was once employed as a bleaching agent and a food preservative.
Q4. What role does boron play in cell phones?
Solution: Borides operate as coatings on metals, protecting them from corrosion and weathering. Many smartphones employ boron because it can produce a protective layer on the metal. Borates are good semiconductor materials and help to prevent corrosion.



